QUESTION IMAGE
Question
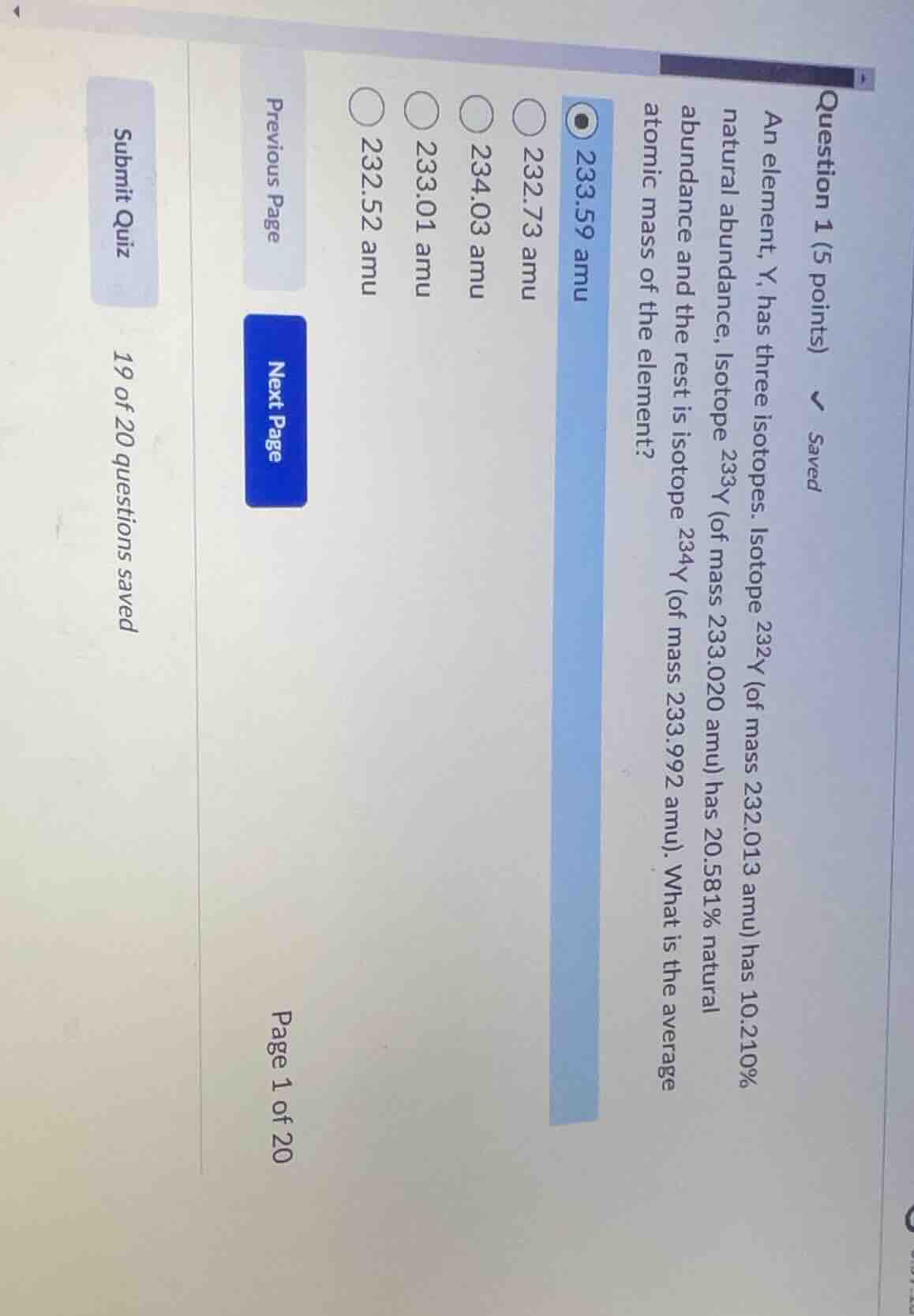
question 1 (5 points) an element, y, has three isotopes. isotope (^{232}\text{y}) (of mass 232.013 amu) has 10.210% natural abundance, isotope (^{233}\text{y}) (of mass 233.020 amu) has 20.581% natural abundance and the rest is isotope (^{234}\text{y}) (of mass 233.992 amu). what is the average atomic mass of the element? (\bigcirc) 233.59 amu (\bigcirc) 232.73 amu (\bigcirc) 234.03 amu (\bigcirc) 233.01 amu (\bigcirc) 232.52 amu
Step1: Find abundance of ²³⁴Y
First, calculate the abundance of the third isotope (²³⁴Y). The total abundance should be 100%, so:
Abundance of ²³⁴Y = \( 100\% - 10.210\% - 20.581\% = 69.209\% \)
Step2: Convert abundances to decimals
Convert each abundance to a decimal (divide by 100):
- Abundance of ²³²Y: \( 0.10210 \)
- Abundance of ²³³Y: \( 0.20581 \)
- Abundance of ²³⁴Y: \( 0.69209 \)
Step3: Calculate weighted average
The formula for average atomic mass is \( \sum (\text{isotope mass} \times \text{abundance}) \).
\[
\]
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
233.59 amu