QUESTION IMAGE
Question
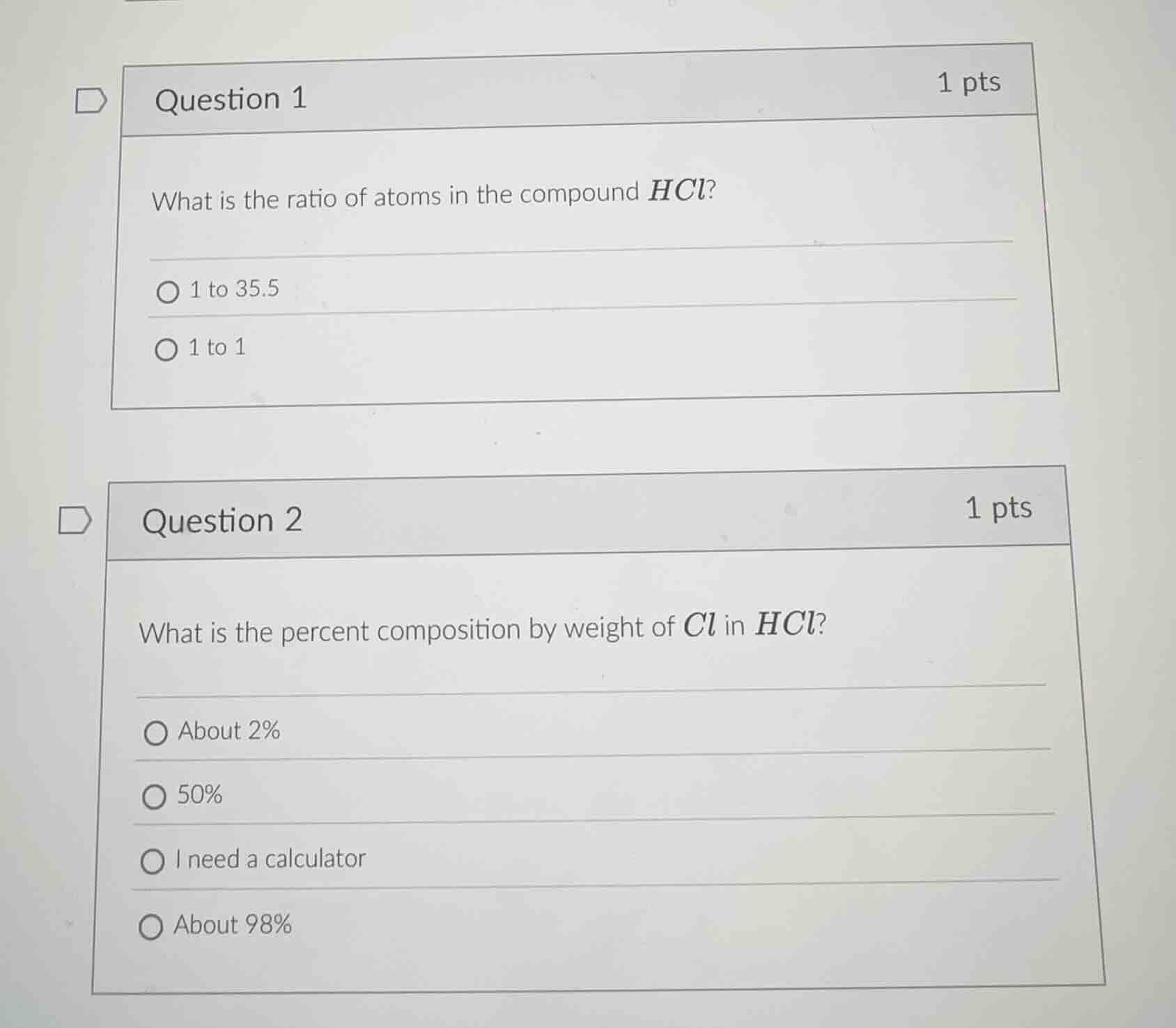
question 1
1 pts
what is the ratio of atoms in the compound hcl?
1 to 35.5
1 to 1
question 2
1 pts
what is the percent composition by weight of cl in hcl?
about 2%
50%
i need a calculator
about 98%
Question 1
In the compound \(HCl\) (hydrochloric acid), the chemical formula shows one hydrogen (\(H\)) atom and one chlorine (\(Cl\)) atom. So the ratio of \(H\) to \(Cl\) atoms is \(1:1\). The option "1 to 35.5" refers to the mass ratio (since the atomic mass of \(H\) is approximately 1 and \(Cl\) is approximately 35.5), not the atom ratio.
Step 1: Find molar masses
The molar mass of \(H\) is approximately \(1\space g/mol\) and the molar mass of \(Cl\) is approximately \(35.5\space g/mol\). The molar mass of \(HCl\) is \(1 + 35.5=36.5\space g/mol\).
Step 2: Calculate percent composition of \(Cl\)
The formula for percent composition of an element in a compound is \(\frac{\text{Molar mass of element in compound}}{\text{Molar mass of compound}}\times100\%\). For \(Cl\) in \(HCl\), it is \(\frac{35.5}{36.5}\times100\%\approx97.26\%\), which is about \(98\%\) (when rounded or approximated). The other options are incorrect: "About 2%" is too low, "50%" is incorrect as the masses of \(H\) and \(Cl\) are not equal, and "I need a calculator" is not an answer but a statement.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1 to 1