QUESTION IMAGE
Question
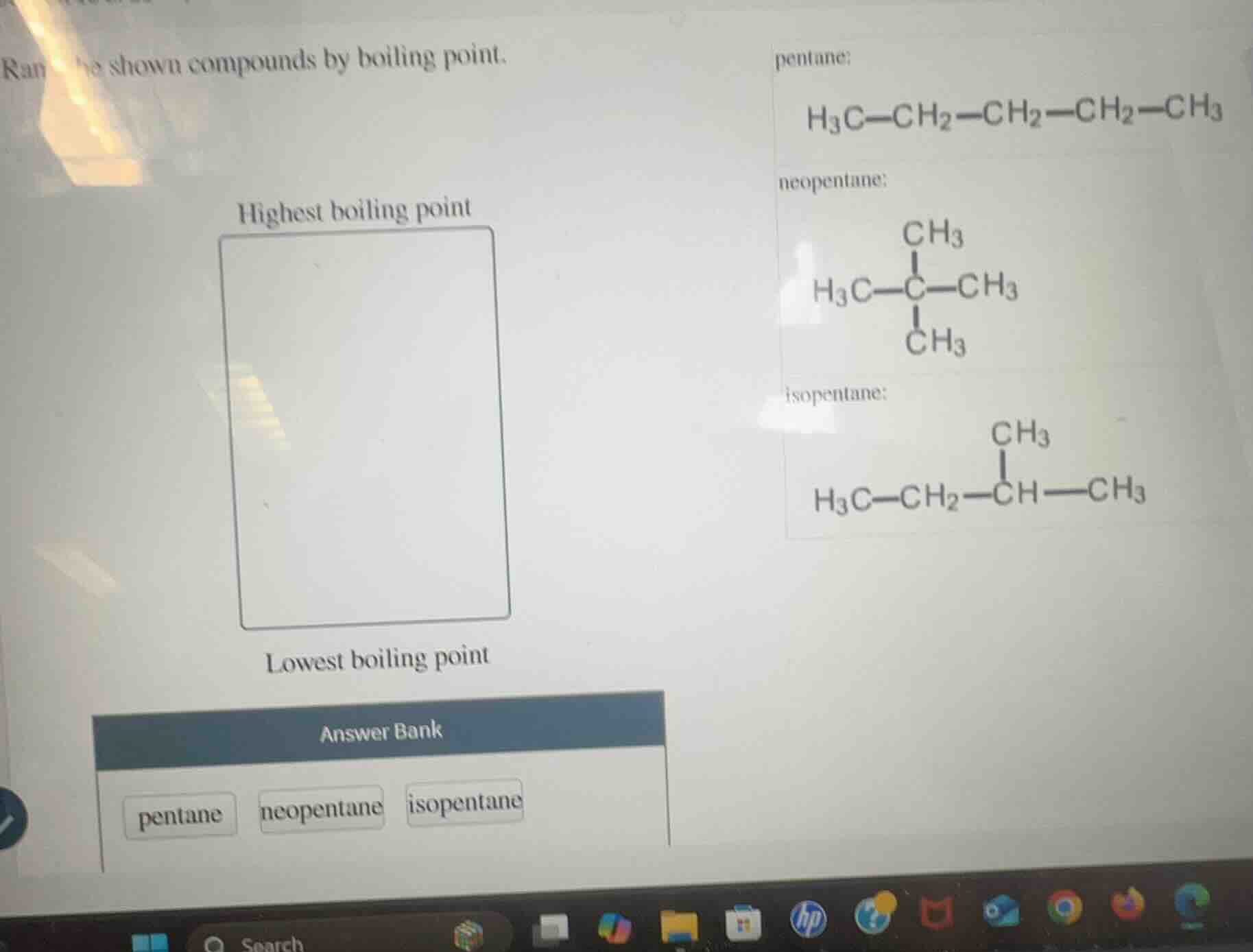
rank the shown compounds by boiling point.
highest boiling point
lowest boiling point
answer bank
pentane
neopentane
isopentane
pentane:
h₃c—ch₂—ch₂—ch₂—ch₃
neopentane:
ch₃
|
h₃c—c—ch₃
|
ch₃
isopentane:
ch₃
|
h₃c—ch₂—ch—ch₃
Boiling points of alkane isomers depend on molecular surface area; more linear molecules have stronger London dispersion forces, higher boiling points. Pentane is linear, isopentane has one branch, neopentane has the most branches (most compact, smallest surface area).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Highest boiling point: pentane ($\text{H}_3\text{C-CH}_2\text{-CH}_2\text{-CH}_2\text{-CH}_3$)
Middle boiling point: isopentane ($\text{H}_3\text{C-CH}_2\text{-CH(CH}_3\text{)-CH}_3$)
Lowest boiling point: neopentane ($\text{(CH}_3\text{)}_4\text{C}$)
Ordered from highest to lowest: pentane > isopentane > neopentane