QUESTION IMAGE
Question
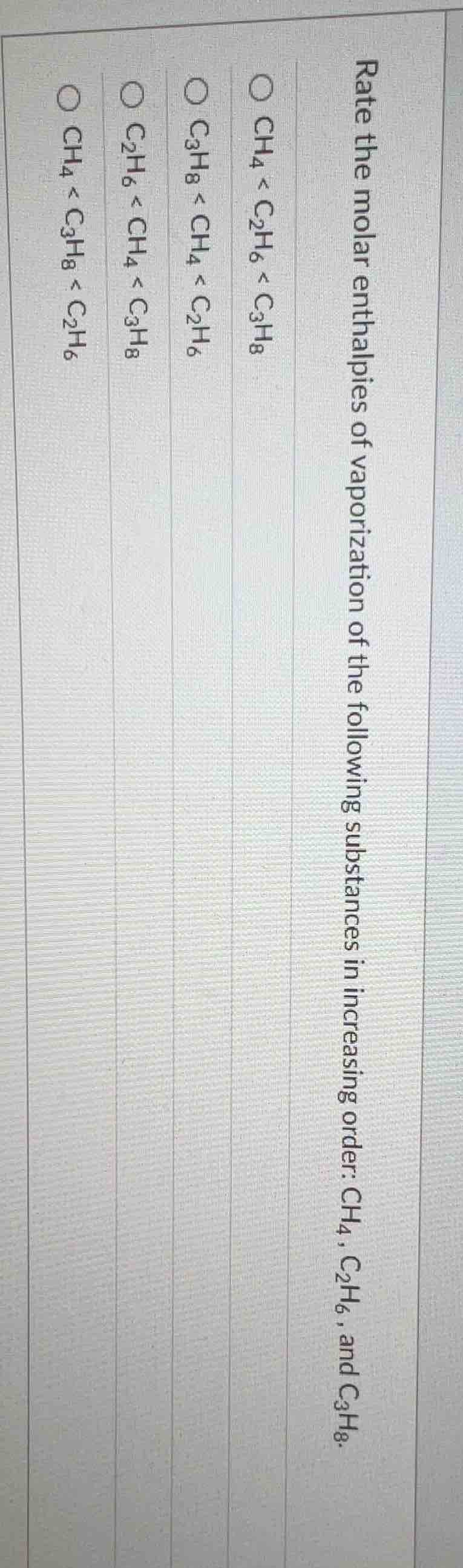
rate the molar enthalpies of vaporization of the following substances in increasing order: ch₄, c₂h₆, and c₃h₈.\
\bigcirc ch₄ < c₂h₆ < c₃h₈\
\bigcirc c₃h₈ < ch₄ < c₂h₆\
\bigcirc c₂h₆ < ch₄ < c₃h₈\
\bigcirc ch₄ < c₃h₈ < c₂h₆
Step1: Recall intermolecular forces
Molar enthalpy of vaporization depends on intermolecular forces. For alkanes (CH₄, C₂H₆, C₃H₈), they are nonpolar, so only London dispersion forces (LDF) act. LDF strength increases with molar mass (or number of electrons, which correlates with molar mass here).
Step2: Compare molar masses
Molar mass of CH₄: \(12 + 4\times1 = 16\) g/mol.
Molar mass of C₂H₆: \(2\times12 + 6\times1 = 30\) g/mol.
Molar mass of C₃H₈: \(3\times12 + 8\times1 = 44\) g/mol.
As molar mass increases, LDF becomes stronger, so more energy (higher molar enthalpy of vaporization) is needed to vaporize. Thus, the order of increasing molar enthalpy of vaporization is CH₄ < C₂H₆ < C₃H₈.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. CH₄ < C₂H₆ < C₃H₈