QUESTION IMAGE
Question
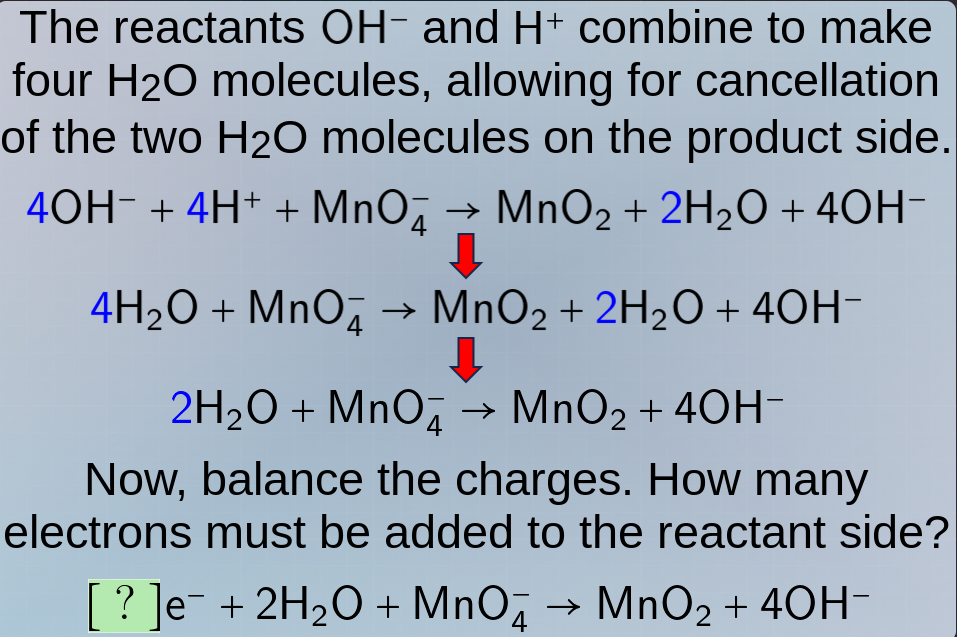
the reactants oh⁻ and h⁺ combine to make four h₂o molecules, allowing for cancellation of the two h₂o molecules on the product side.
4oh⁻ + 4h⁺ + mno₄⁻ → mno₂ + 2h₂o + 4oh⁻
4h₂o + mno₄⁻ → mno₂ + 2h₂o + 4oh⁻
2h₂o + mno₄⁻ → mno₂ + 4oh⁻
now, balance the charges. how many electrons must be added to the reactant side?
?e⁻ + 2h₂o + mno₄⁻ → mno₂ + 4oh⁻
Step1: Calculate charge on reactant side
Reactants: \(2H_2O\) (neutral, charge 0), \(MnO_4^-\) (charge -1). Total reactant charge: \(0 + (-1)= -1\)
Step2: Calculate charge on product side
Products: \(MnO_2\) (neutral, charge 0), \(4OH^-\) (each \(OH^-\) is -1, so \(4\times(-1)= -4\)). Total product charge: \(0 + (-4)= -4\)
Step3: Determine electrons needed
Let \(x\) be electrons (each \(e^-\) has -1 charge). Reactant charge with electrons: \(-1 + (-x)\) (since electrons are added to reactant side, their charge is -1 each). This should equal product charge (-4):
\(-1 - x = -4\)
Solve for \(x\): \(-x = -4 + 1 = -3\) → \(x = 3\) (since charge of electron is -1, adding 3 electrons (total charge -3) to reactant side makes reactant charge \(-1 + (-3)= -4\), equal to product charge)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
3