QUESTION IMAGE
Question
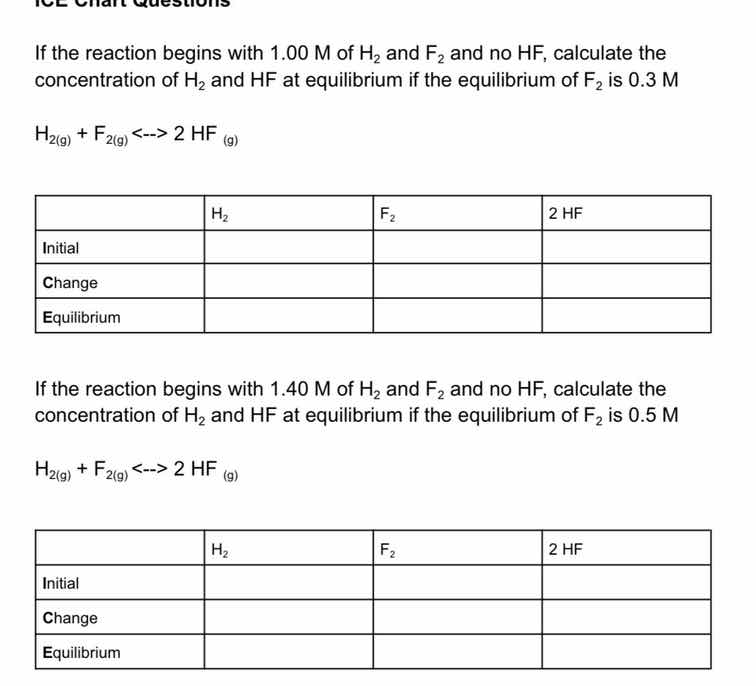
if the reaction begins with 1.00 m of h₂ and f₂ and no hf, calculate the concentration of h₂ and hf at equilibrium if the equilibrium of f₂ is 0.3 m
h₂(g) + f₂(g) <--> 2 hf (g)
| h₂ | f₂ | 2 hf | |
| initial | |||
| change | |||
| equilibrium |
if the reaction begins with 1.40 m of h₂ and f₂ and no hf, calculate the concentration of h₂ and hf at equilibrium if the equilibrium of f₂ is 0.5 m
h₂(g) + f₂(g) <--> 2 hf (g)
| h₂ | f₂ | 2 hf | |
| initial | |||
| change | |||
| equilibrium |
---
First Problem:
Step1: Fill initial concentrations
Initial: $[\text{H}_2] = 1.00\ \text{M}$, $[\text{F}_2] = 1.00\ \text{M}$, $[\text{HF}] = 0\ \text{M}$
Step2: Find change in $\text{F}_2$
Change in $\text{F}_2$: $1.00 - 0.3 = 0.7\ \text{M}$
Step3: Relate changes to stoichiometry
$\Delta [\text{H}_2] = -0.7\ \text{M}$, $\Delta [\text{HF}] = +2\times0.7 = 1.4\ \text{M}$
Step4: Calculate equilibrium values
$[\text{H}_2]_{\text{eq}} = 1.00 - 0.7 = 0.3\ \text{M}$
$[\text{HF}]_{\text{eq}} = 0 + 1.4 = 1.4\ \text{M}$
---
Second Problem:
Step1: Fill initial concentrations
Initial: $[\text{H}_2] = 1.40\ \text{M}$, $[\text{F}_2] = 1.40\ \text{M}$, $[\text{HF}] = 0\ \text{M}$
Step2: Find change in $\text{F}_2$
Change in $\text{F}_2$: $1.40 - 0.5 = 0.9\ \text{M}$
Step3: Relate changes to stoichiometry
$\Delta [\text{H}_2] = -0.9\ \text{M}$, $\Delta [\text{HF}] = +2\times0.9 = 1.8\ \text{M}$
Step4: Calculate equilibrium values
$[\text{H}_2]_{\text{eq}} = 1.40 - 0.9 = 0.5\ \text{M}$
$[\text{HF}]_{\text{eq}} = 0 + 1.8 = 1.8\ \text{M}$
---
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First ICE Table and Results:
| $\text{H}_2$ | $\text{F}_2$ | $2\text{HF}$ | |
|---|---|---|---|
| Change | $-0.7\ \text{M}$ | $-0.7\ \text{M}$ | $+1.4\ \text{M}$ |
| Equilibrium | $0.3\ \text{M}$ | $0.3\ \text{M}$ | $1.4\ \text{M}$ |
Equilibrium $[\text{H}_2] = 0.3\ \text{M}$, Equilibrium $[\text{HF}] = 1.4\ \text{M}$
Second ICE Table and Results:
| $\text{H}_2$ | $\text{F}_2$ | $2\text{HF}$ | |
|---|---|---|---|
| Change | $-0.9\ \text{M}$ | $-0.9\ \text{M}$ | $+1.8\ \text{M}$ |
| Equilibrium | $0.5\ \text{M}$ | $0.5\ \text{M}$ | $1.8\ \text{M}$ |
Equilibrium $[\text{H}_2] = 0.5\ \text{M}$, Equilibrium $[\text{HF}] = 1.8\ \text{M}$