QUESTION IMAGE
Question
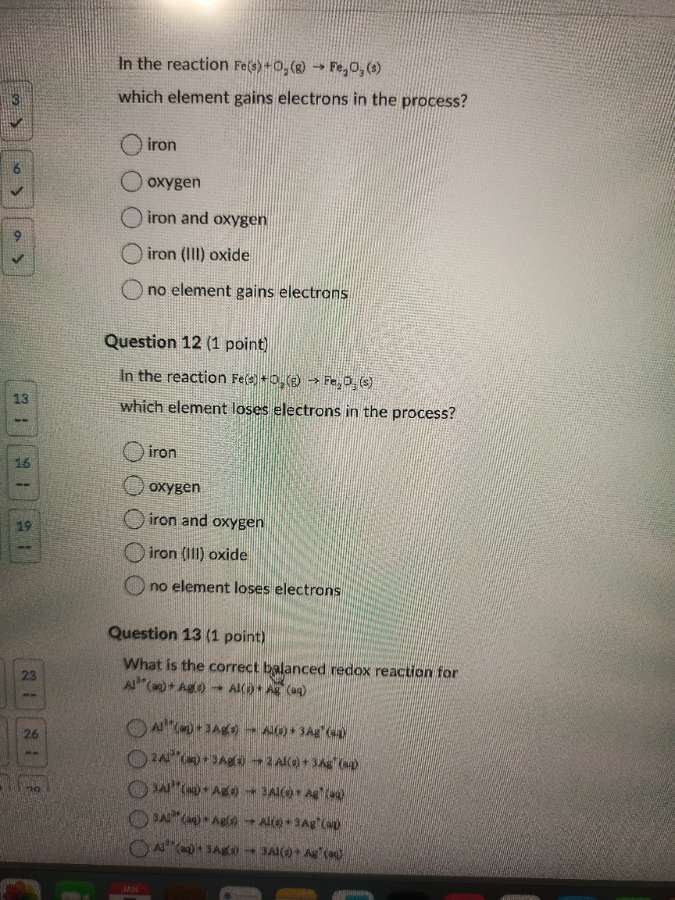
in the reaction fe(s) + o₂(g) → fe₂o₃(s) which element gains electrons in the process?
○ iron
○ oxygen
○ iron and oxygen
○ iron (iii) oxide
○ no element gains electrons
question 12 (1 point)
in the reaction fe(s) + o₂(g) → fe₂o₃(s) which element loses electrons in the process?
○ iron
○ oxygen
○ iron and oxygen
○ iron (iii) oxide
○ no element loses electrons
question 13 (1 point)
what is the correct balanced redox reaction for al³⁺(aq) + ag(s) → al(s) + ag⁺(aq)
○ al³⁺(aq) + 3ag(s) → al(s) + 3ag⁺(aq)
○ 2al³⁺(aq) + 3ag(s) → 2al(s) + 3ag⁺(aq)
○ 3al³⁺(aq) + ag(s) → 3al(s) + ag⁺(aq)
○ 2al³⁺(aq) + ag(s) → al(s) + 3ag⁺(aq)
○ al³⁺(aq) + 3ag(s) → 3al(s) + ag⁺(aq)
Question (First part: Which element gains electrons in $\ce{Fe(s) + O_{2}(g) -> Fe_{2}O_{3}(s)}$)
To determine which element gains electrons, we analyze oxidation states. In $\ce{O_{2}(g)}$, oxygen has an oxidation state of 0. In $\ce{Fe_{2}O_{3}}$, oxygen has an oxidation state of -2 (since oxygen in oxides is typically -2, and the sum of oxidation states in a compound is zero: for $\ce{Fe_{2}O_{3}}$, let Fe be $x$, so $2x + 3(-2)=0 \implies x = +3$). A decrease in oxidation state means gain of electrons (reduction). Oxygen's oxidation state goes from 0 to -2, so it gains electrons. Iron in $\ce{Fe(s)}$ has an oxidation state of 0, and in $\ce{Fe_{2}O_{3}}$ it is +3, so iron loses electrons (oxidation).
As analyzed earlier, iron in $\ce{Fe(s)}$ has an oxidation state of 0. In $\ce{Fe_{2}O_{3}}$, iron has an oxidation state of +3. An increase in oxidation state means loss of electrons (oxidation). Oxygen's oxidation state decreases (from 0 to -2), so it gains electrons. Thus, iron loses electrons.
Step 1: Identify oxidation and reduction half - reactions
- Oxidation (Ag): $\ce{Ag(s) -> Ag^{+}(aq) + e^{-}}$ (Ag goes from 0 to +1, loses 1 electron)
- Reduction (Al): $\ce{Al^{3+}(aq) + 3e^{-} -> Al(s)}$ (Al goes from +3 to 0, gains 3 electrons)
Step 2: Balance electrons
To balance the electrons, we need to multiply the oxidation half - reaction by 3 so that the number of electrons lost (3) equals the number of electrons gained (3).
- Oxidation (multiplied by 3): $\ce{3Ag(s) -> 3Ag^{+}(aq) + 3e^{-}}$
- Reduction: $\ce{Al^{3+}(aq) + 3e^{-} -> Al(s)}$
Step 3: Add the half - reactions
Add the two balanced half - reactions together:
$\ce{Al^{3+}(aq) + 3Ag(s) + 3e^{-} -> Al(s) + 3Ag^{+}(aq) + 3e^{-}}$
The electrons ($\ce{3e^{-}}$) cancel out, giving the balanced reaction: $\ce{Al^{3+}(aq) + 3Ag(s) -> Al(s) + 3Ag^{+}(aq)}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. oxygen