QUESTION IMAGE
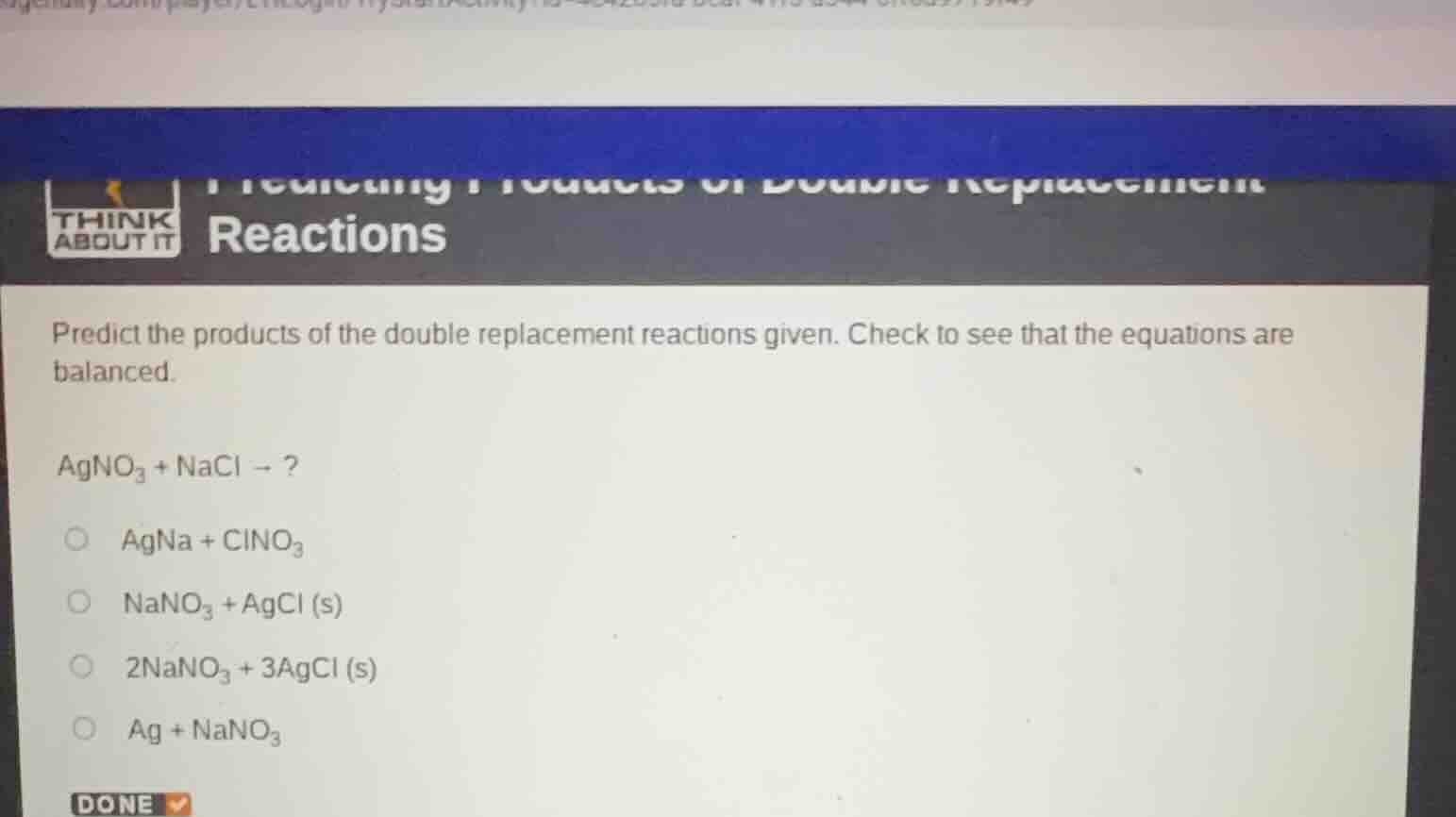
Question
reactions
predict the products of the double replacement reactions given. check to see that the equations are balanced.
$\text{agno}_3 + \text{nacl}
ightarrow ?$
$\text{agna} + \text{clno}_3$
$\text{nano}_3 + \text{agcl (s)}$
$2\text{nano}_3 + 3\text{agcl (s)}$
$\text{ag} + \text{nano}_3$
Step1: Identify double replacement pairs
In double replacement, cations/anions swap: $\text{Ag}^+$ pairs with $\text{Cl}^-$, $\text{Na}^+$ pairs with $\text{NO}_3^-$.
Step2: Form valid products
$\text{AgCl}$ is an insoluble solid (precipitate), $\text{NaNO}_3$ is soluble.
Step3: Balance the equation
Count atoms: 1 Ag, 1 N, 3 O, 1 Na, 1 Cl on both sides. Equation is balanced: $\text{AgNO}_3 + \text{NaCl}
ightarrow \text{NaNO}_3 + \text{AgCl (s)}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\text{NaNO}_3 + \text{AgCl (s)}$