QUESTION IMAGE
Question
reactions
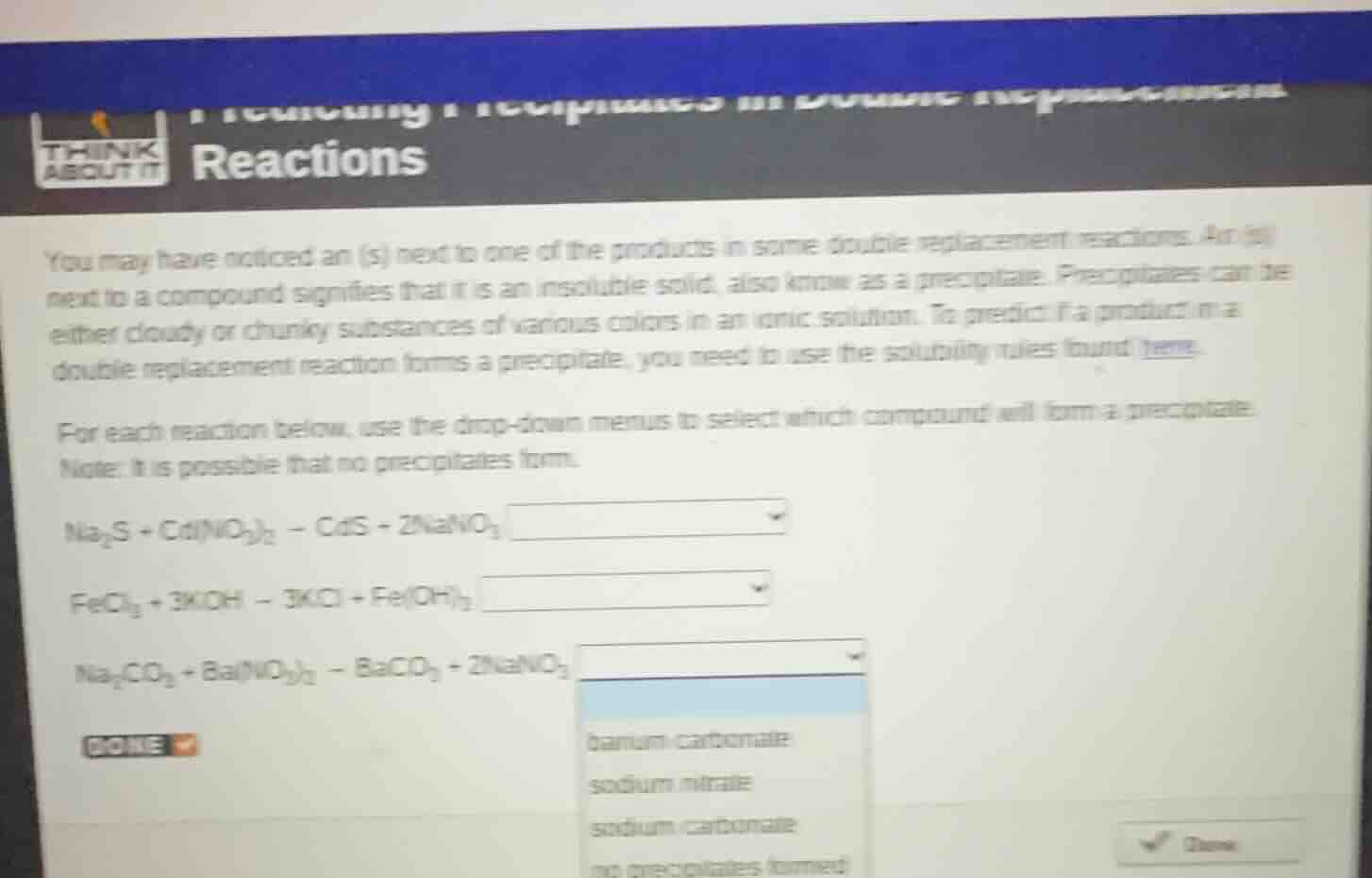
you may have noticed an (s) next to one of the products in some double replacement reactions. an (s)
next to a compound signifies that it is an insoluble solid, also know as a precipitate. precipitates can be
either cloudy or chunky substances of various colors in an ionic solution. to predict if a product in a
double replacement reaction forms a precipitate, you need to use the solubility rules found here.
for each reaction below, use the drop-down menus to select which compound will form a precipitate.
note: it is possible that no precipitates form.
$ce{na_{2}s + cd(no_{3})_{2} -> cds + 2nano_{3}}$
$ce{fecl_{3} + 3koh -> 3kcl + fe(oh)_{3}}$
$ce{na_{2}co_{3} + ba(no_{3})_{2} -> baco_{3} + 2nano_{3}}$
barium carbonate
sodium nitrate
sodium carbonate
no precipitates formed
Step1: Apply solubility rules to reaction 1
All nitrates are soluble; sulfides of Cd are insoluble. So $\text{CdS}$ is the precipitate.
Step2: Apply solubility rules to reaction 2
All chlorides (with K⁺) are soluble; hydroxides of Fe³⁺ are insoluble. So $\text{Fe(OH)}_3$ is the precipitate.
Step3: Apply solubility rules to reaction 3
All nitrates are soluble; carbonates of Ba²⁺ are insoluble. So $\text{BaCO}_3$ (barium carbonate) is the precipitate.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{CdS}$
- $\text{Fe(OH)}_3$
- barium carbonate