QUESTION IMAGE
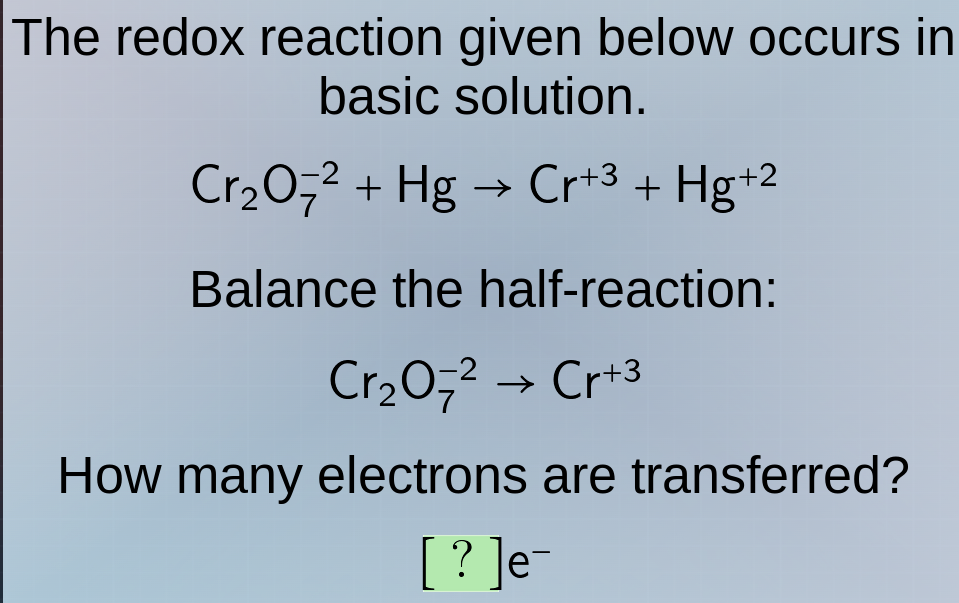
Question
the redox reaction given below occurs in basic solution.\\(\ce{cr2o7^{-2} + hg -> cr^{+3} + hg^{+2}}\\)\
balance the half - reaction:\\(\ce{cr2o7^{-2} -> cr^{+3}}\\)\
how many electrons are transferred?\\(\ce{?e^-}\\)
Step1: Balance Cr atoms
In the half - reaction \( \text{Cr}_2\text{O}_7^{2 - }\to\text{Cr}^{3 + } \), we have 2 Cr atoms on the left and 1 on the right. So we multiply \( \text{Cr}^{3+} \) by 2:
\( \text{Cr}_2\text{O}_7^{2 - }\to2\text{Cr}^{3 + } \)
Step2: Balance O atoms by adding \( \text{H}_2\text{O} \)
There are 7 O atoms on the left. So we add 7 \( \text{H}_2\text{O} \) molecules to the right to balance O:
\( \text{Cr}_2\text{O}_7^{2 - }\to2\text{Cr}^{3 + }+7\text{H}_2\text{O} \)
Step3: Balance H atoms by adding \( \text{H}^+ \) (then adjust for basic solution later, but for charge balance we can use \( \text{H}^+ \) first)
There are 14 H atoms in \( 7\text{H}_2\text{O} \) on the right. So we add 14 \( \text{H}^+ \) to the left:
\( 14\text{H}^++\text{Cr}_2\text{O}_7^{2 - }\to2\text{Cr}^{3 + }+7\text{H}_2\text{O} \)
Step4: Balance charge
The charge on the left side: \( 14(+1)+(- 2)= + 12 \)
The charge on the right side: \( 2(+3)=+6 \)
The difference in charge is \( + 12-(+6) = + 6 \). Since the reaction is a reduction (Cr is going from +6 in \( \text{Cr}_2\text{O}_7^{2 - } \) to +3 in \( \text{Cr}^{3+} \)), we need to gain electrons (each electron has a - 1 charge). Let the number of electrons be \( n \). Then:
\( + 12-n=+6 \)
Solving for \( n \), we get \( n = 6 \). Alternatively, we can calculate the oxidation state change of Cr. In \( \text{Cr}_2\text{O}_7^{2 - } \), let the oxidation state of Cr be \( x \). Then \( 2x + 7(-2)=-2\), so \( 2x-14 = - 2\), \( 2x=12 \), \( x = + 6 \). Each Cr atom changes from +6 to +3, a change of - 3 per Cr atom. Since there are 2 Cr atoms, the total change in oxidation state is \( 2\times(6 - 3)=6 \). So 6 electrons are gained (transferred) in this half - reaction.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
6