QUESTION IMAGE
Question
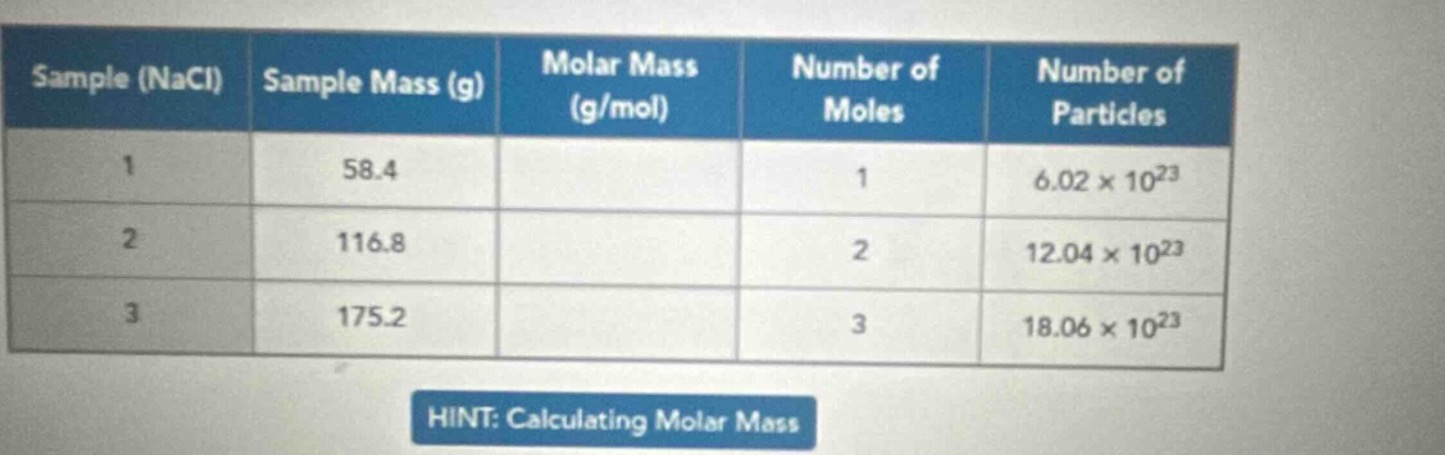
sample (nacl) | sample mass (g) | molar mass (g/mol) | number of moles | number of particles
1 | 58.4 | | 1 | 6.02×10²³
2 | 116.8 | | 2 | 12.04×10²³
3 | 175.2 | | 3 | 18.06×10²³
hint: calculating molar mass
To determine the molar mass of NaCl, we use the formula for calculating molar mass:
Step 1: Recall the formula for molar mass
The molar mass ($M$) is calculated as the ratio of the sample mass ($m$) to the number of moles ($n$):
$$M = \frac{m}{n}$$
Step 2: Calculate molar mass for Sample 1
For Sample 1:
- Sample mass ($m$) = 58.4 g
- Number of moles ($n$) = 1 mol
Substitute into the formula:
$$M = \frac{58.4\ \text{g}}{1\ \text{mol}} = 58.4\ \text{g/mol}$$
Step 3: Verify with Sample 2
For Sample 2:
- Sample mass ($m$) = 116.8 g
- Number of moles ($n$) = 2 mol
Substitute into the formula:
$$M = \frac{116.8\ \text{g}}{2\ \text{mol}} = 58.4\ \text{g/mol}$$
Step 4: Verify with Sample 3
For Sample 3:
- Sample mass ($m$) = 175.2 g
- Number of moles ($n$) = 3 mol
Substitute into the formula:
$$M = \frac{175.2\ \text{g}}{3\ \text{mol}} = 58.4\ \text{g/mol}$$
All samples confirm that the molar mass of NaCl is consistent.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The molar mass of NaCl is $\boldsymbol{58.4\ \text{g/mol}}$.