QUESTION IMAGE
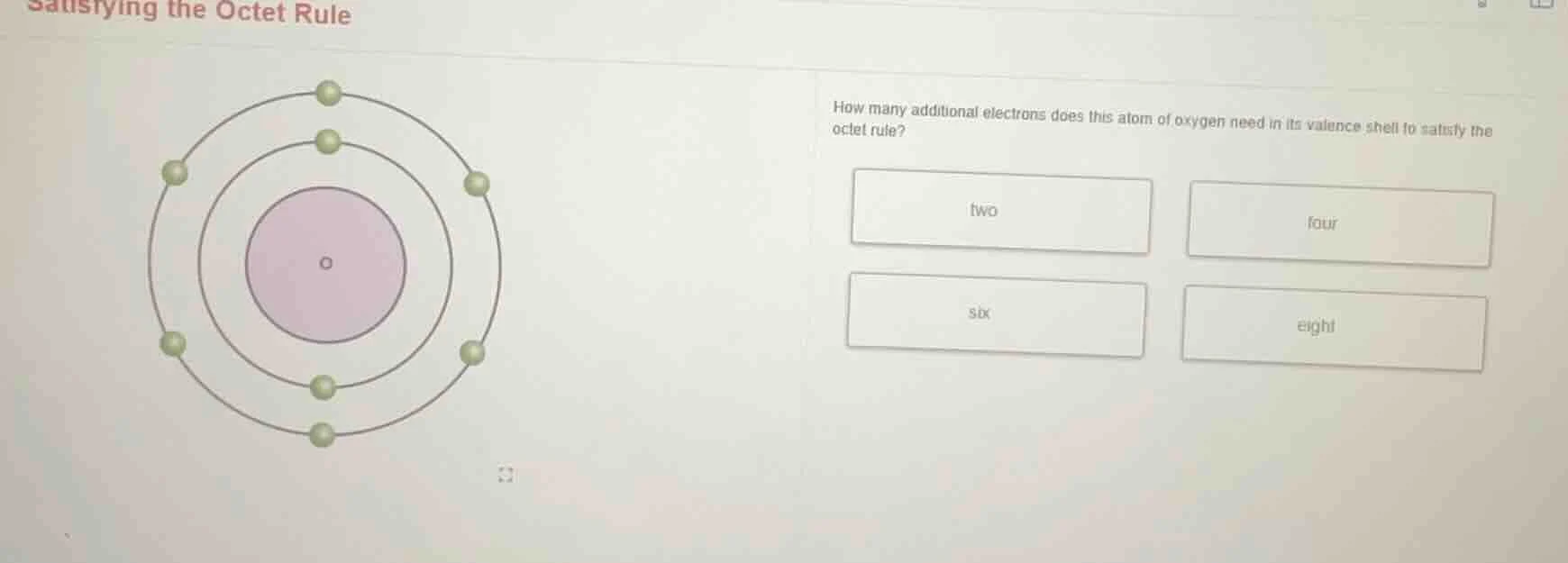
Question
satisfying the octet rule
how many additional electrons does this atom of oxygen need in its valence shell to satisfy the octet rule?
two four
six eight
Step1: Identify valence electrons
Oxygen has 6 valence electrons (from the diagram, the outer shell has 6 electrons).
Step2: Apply octet rule
The octet rule requires 8 valence electrons. Calculate the deficit: $8 - 6 = 2$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
two