QUESTION IMAGE
Question
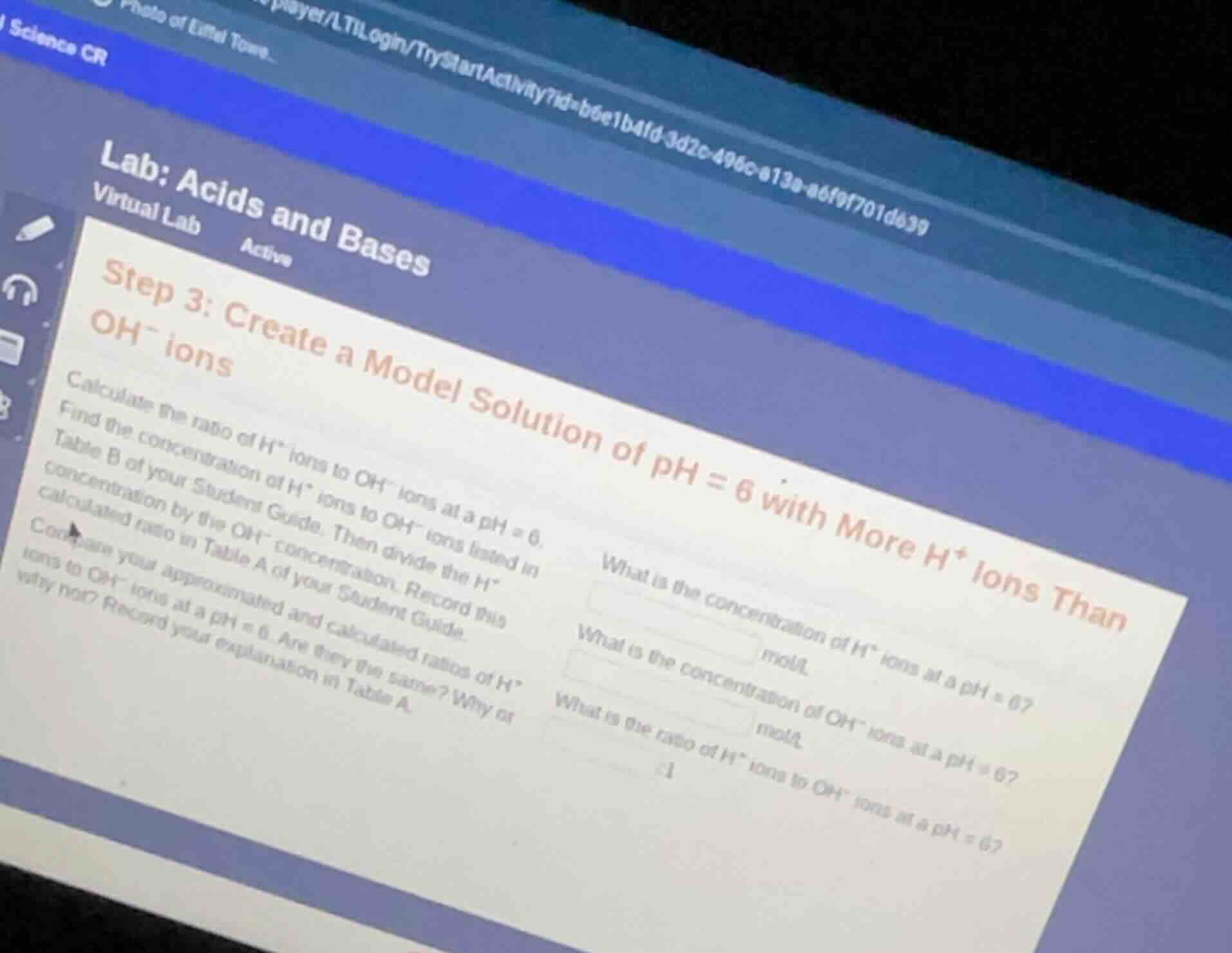
science cr
lab: acids and bases
virtual lab active
step 3: create a model solution of ph = 6 with more h⁺ ions than oh⁻ ions
calculate the ratio of h⁺ ions to oh⁻ ions at a ph = 6.
find the concentration of h⁺ ions to oh⁻ ions listed in table b of your student guide. then divide the h⁺ concentration by the oh⁻ concentration. record this calculated ratio in table a of your student guide.
compare your approximated and calculated ratios of h⁺ ions to oh⁻ ions at a ph = 6. are they the same? why or why not? record your explanation in table a.
what is the concentration of h⁺ ions at a ph = 6? ______ mol/l
what is the concentration of oh⁻ ions at a ph = 6? ______ mol/l
what is the ratio of h⁺ ions to oh⁻ ions at a ph = 6? ______:1
Step1: Calculate $[\text{H}^+]$ from pH
The formula for pH is $\text{pH} = -\log_{10}[\text{H}^+]$. Rearranging gives $[\text{H}^+] = 10^{-\text{pH}}$.
For $\text{pH}=6$:
$[\text{H}^+] = 10^{-6}\ \text{mol/L}$
Step2: Calculate $[\text{OH}^-]$ from $K_w$
At 25°C, $K_w = [\text{H}^+][\text{OH}^-] = 10^{-14}$. Rearranging gives $[\text{OH}^-] = \frac{10^{-14}}{[\text{H}^+]}$.
Substitute $[\text{H}^+]=10^{-6}$:
$[\text{OH}^-] = \frac{10^{-14}}{10^{-6}} = 10^{-8}\ \text{mol/L}$
Step3: Find $\text{H}^+:\text{OH}^-$ ratio
Divide $[\text{H}^+]$ by $[\text{OH}^-]$:
$\frac{[\text{H}^+]}{[\text{OH}^-]} = \frac{10^{-6}}{10^{-8}} = 100$
So the ratio is $100:1$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
What is the concentration of $\text{H}^+$ ions at a pH = 6?
$10^{-6}\ \text{mol/L}$
What is the concentration of $\text{OH}^-$ ions at a pH = 6?
$10^{-8}\ \text{mol/L}$
What is the ratio of $\text{H}^+$ ions to $\text{OH}^-$ ions at a pH = 6?
$100:1$