QUESTION IMAGE
Question
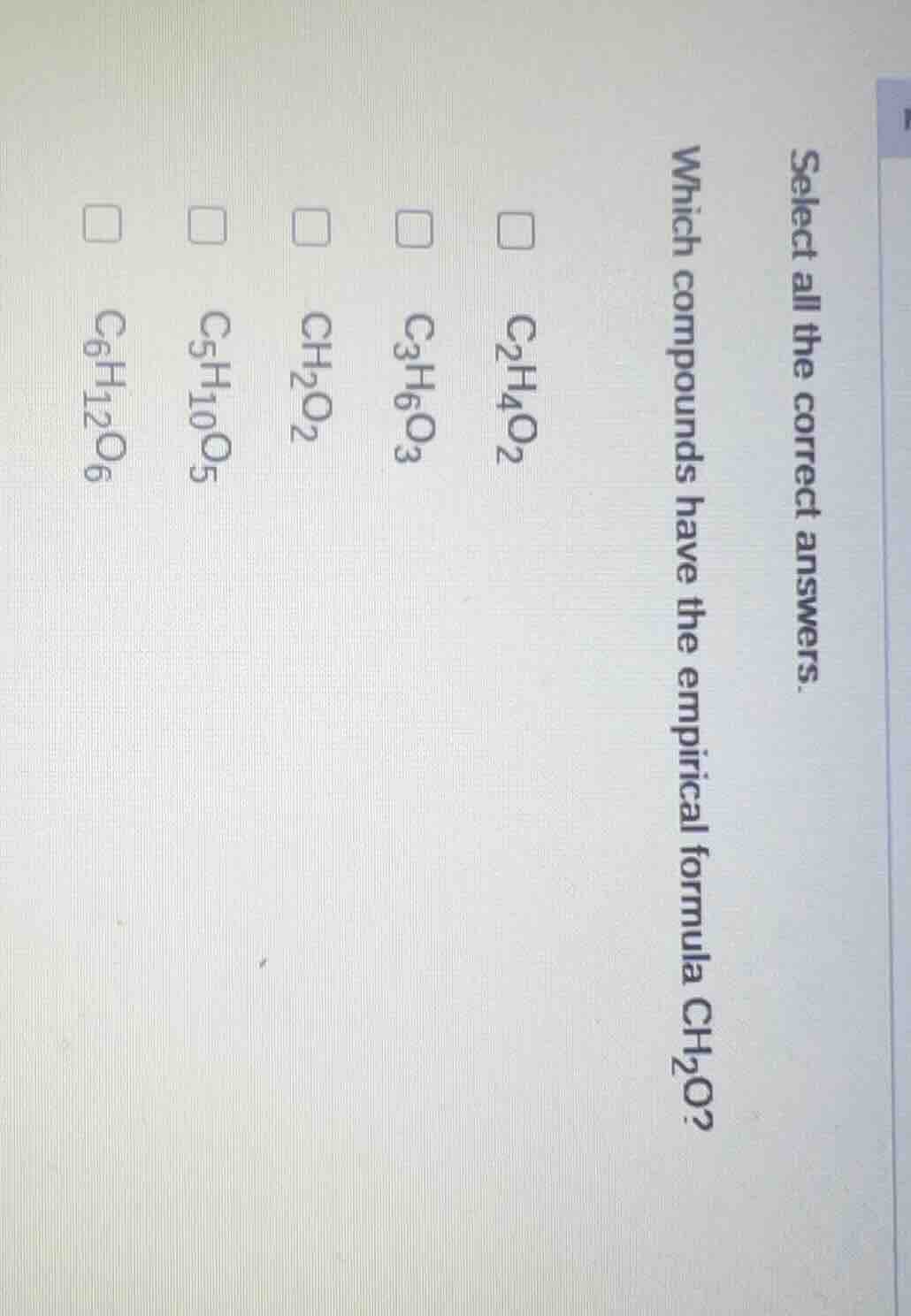
select all the correct answers. which compounds have the empirical formula ch₂o? c₂h₄o₂ c₃h₆o₃ ch₂o₂ c₅h₁₀o₅ c₆h₁₂o₆
To determine which compounds have the empirical formula \( CH_2O \), we need to check if the molecular formula can be simplified to \( CH_2O \) by dividing the subscripts by their greatest common divisor (GCD).
Step 1: Analyze \( C_2H_4O_2 \)
The subscripts are 2, 4, and 2. The GCD of 2, 4, and 2 is 2. Dividing each subscript by 2: \( \frac{2}{2} = 1 \), \( \frac{4}{2} = 2 \), \( \frac{2}{2} = 1 \). So the empirical formula is \( CH_2O \).
Step 2: Analyze \( C_3H_6O_3 \)
The subscripts are 3, 6, and 3. The GCD of 3, 6, and 3 is 3. Dividing each subscript by 3: \( \frac{3}{3} = 1 \), \( \frac{6}{3} = 2 \), \( \frac{3}{3} = 1 \). So the empirical formula is \( CH_2O \).
Step 3: Analyze \( CH_2O_2 \)
The subscripts are 1, 2, and 2. The GCD of 1, 2, and 2 is 1. So the empirical formula is \( CH_2O_2 \), not \( CH_2O \).
Step 4: Analyze \( C_5H_{10}O_5 \)
The subscripts are 5, 10, and 5. The GCD of 5, 10, and 5 is 5. Dividing each subscript by 5: \( \frac{5}{5} = 1 \), \( \frac{10}{5} = 2 \), \( \frac{5}{5} = 1 \). So the empirical formula is \( CH_2O \).
Step 5: Analyze \( C_6H_{12}O_6 \)
The subscripts are 6, 12, and 6. The GCD of 6, 12, and 6 is 6. Dividing each subscript by 6: \( \frac{6}{6} = 1 \), \( \frac{12}{6} = 2 \), \( \frac{6}{6} = 1 \). So the empirical formula is \( CH_2O \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The compounds with the empirical formula \( CH_2O \) are:
- \( C_2H_4O_2 \)
- \( C_3H_6O_3 \)
- \( C_5H_{10}O_5 \)
- \( C_6H_{12}O_6 \)