QUESTION IMAGE
Question
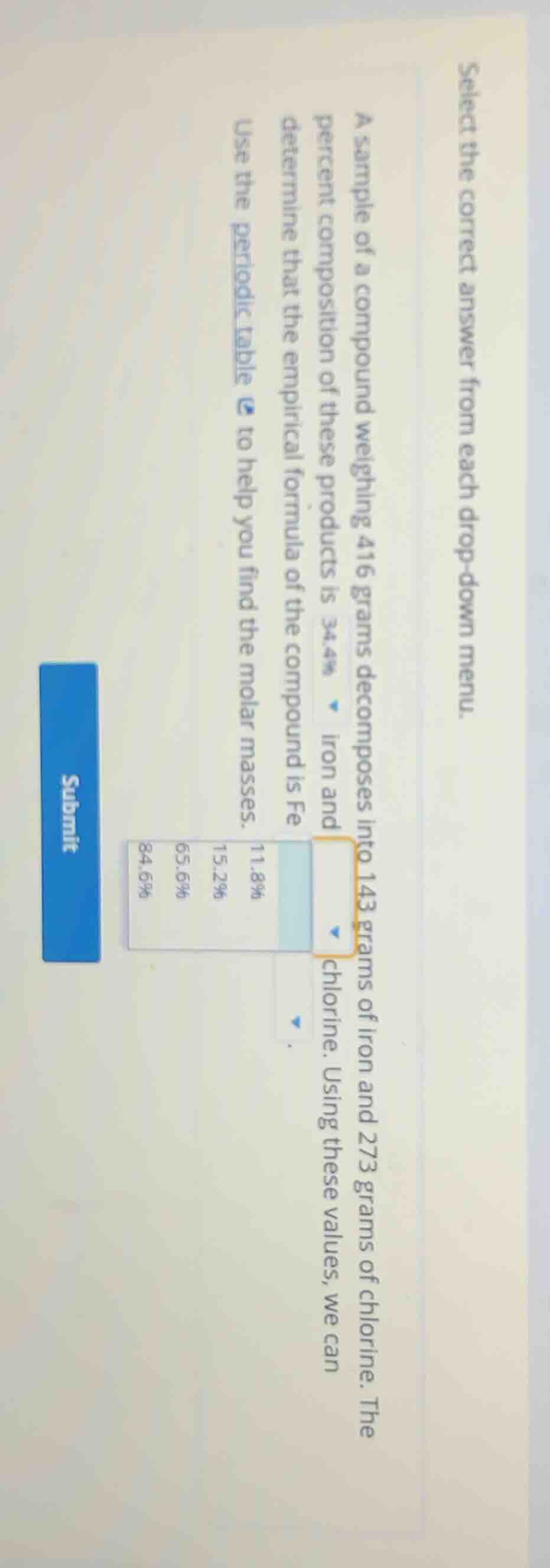
select the correct answer from each drop - down menu. a sample of a compound weighing 416 grams decomposes into 143 grams of iron and 273 grams of chlorine. the percent composition of these products is 34.4% iron and chlorine. using these values, we can determine that the empirical formula of the compound is fe use the periodic table to help you find the molar masses.
Step1: Calculate mass percent of iron
To find the mass percent of iron, use the formula: $\text{Mass Percent} = \frac{\text{Mass of Component}}{\text{Total Mass of Compound}} \times 100\%$. The mass of iron is 143 g, and the total mass of the compound is 416 g. So, $\text{Mass Percent of Fe} = \frac{143}{416} \times 100\% \approx 34.4\%$ (which matches the given value).
Step2: Calculate mass percent of chlorine
The mass of chlorine is 273 g. Using the same formula: $\text{Mass Percent of Cl} = \frac{273}{416} \times 100\% \approx 65.6\%$.
Step3: Determine moles of each element
- Molar mass of Fe: 55.85 g/mol. Moles of Fe: $\frac{143\ \text{g}}{55.85\ \text{g/mol}} \approx 2.56\ \text{mol}$.
- Molar mass of Cl: 35.45 g/mol. Moles of Cl: $\frac{273\ \text{g}}{35.45\ \text{g/mol}} \approx 7.70\ \text{mol}$.
Step4: Find the mole ratio
Divide moles of each by the smallest number of moles (2.56 mol for Fe):
- Ratio for Fe: $\frac{2.56}{2.56} = 1$.
- Ratio for Cl: $\frac{7.70}{2.56} \approx 3$.
So the empirical formula is $\text{FeCl}_3$. The mass percent of chlorine is 65.6%, so the correct percentage for chlorine is 65.6%.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The mass percent of chlorine is 65.6%, so the correct option from the dropdown (for chlorine's percent) is 65.6%. The empirical formula is $\text{FeCl}_3$, so the subscripts would be Fe (1) and Cl (3), but based on the percent calculation for chlorine, the answer for the chlorine percent is 65.6%.