QUESTION IMAGE
Question
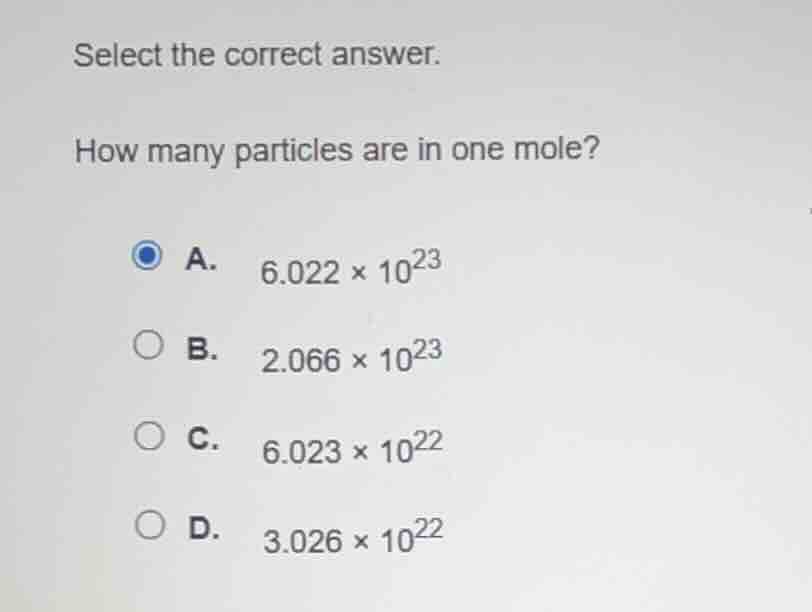
select the correct answer. how many particles are in one mole? a. $6.022 × 10^{23}$ b. $2.066 × 10^{23}$ c. $6.023 × 10^{22}$ d. $3.026 × 10^{22}$
Brief Explanations
This is a foundational concept in chemistry, where Avogadro's number defines the number of particles (atoms, molecules, ions, etc.) in one mole of a substance, which is $6.022 \times 10^{23}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. $6.022 \times 10^{23}$