QUESTION IMAGE
Question
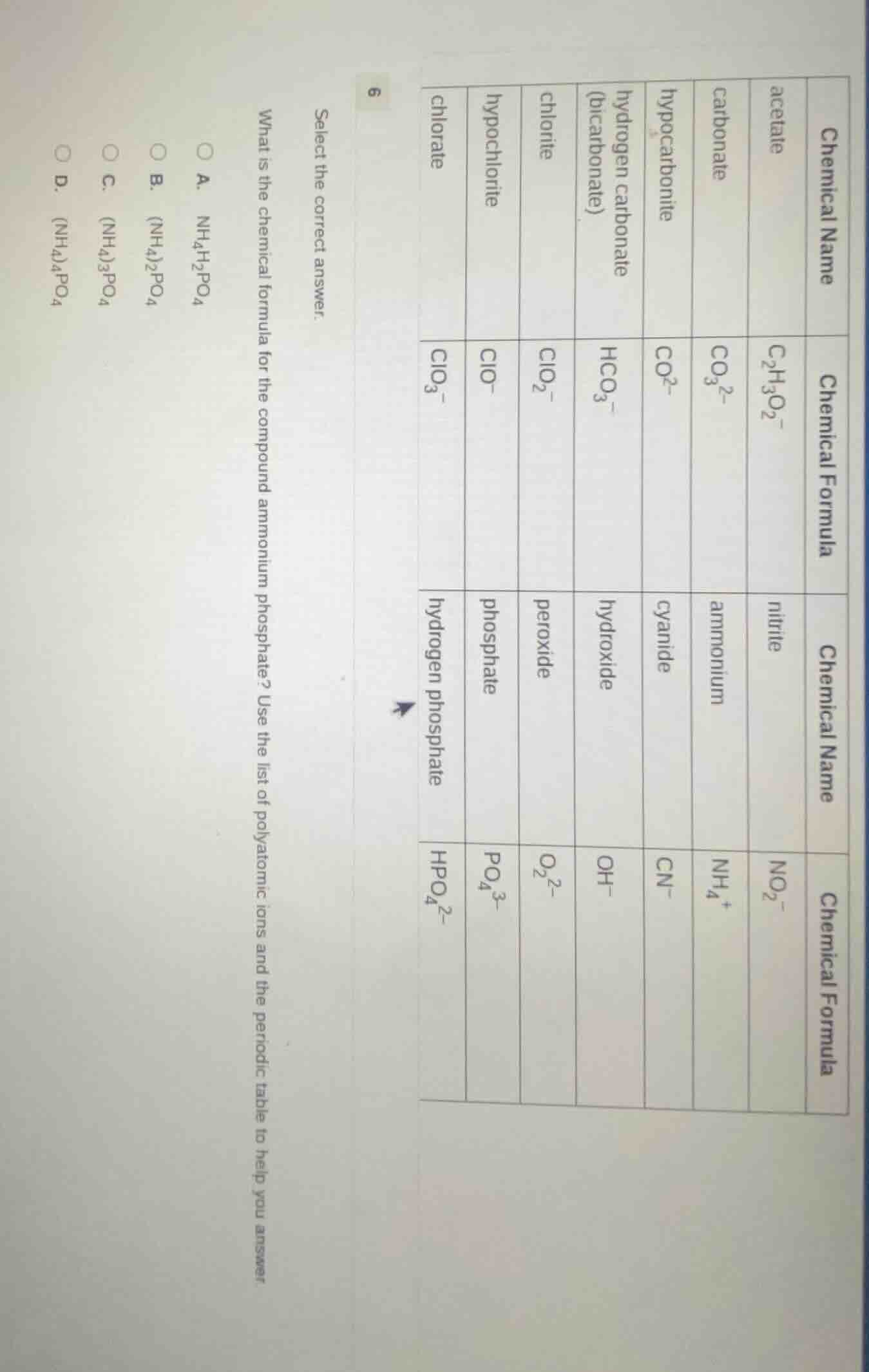
select the correct answer.
what is the chemical formula for the compound ammonium phosphate? use the list of polyatomic ions and the periodic table to help you answer.
a. $\ce{nh4h2po4}$
b. $\ce{(nh4)2po4}$
c. $\ce{(nh4)3po4}$
d. $\ce{(nh4)4po4}$
| chemical name | chemical formula | chemical name | chemical formula |
|---|---|---|---|
| carbonate | $\ce{co3^{2-}}$ | ammonium | $\ce{nh4^+}$ |
| hypocarbonite | $\ce{co2^-}$ | cyanide | $\ce{cn^-}$ |
| hydrogen carbonate (bicarbonate) | $\ce{hco3^-}$ | hydroxide | $\ce{oh^-}$ |
| chlorite | $\ce{clo2^-}$ | peroxide | $\ce{o2^{2-}}$ |
| hypochlorite | $\ce{clo^-}$ | phosphate | $\ce{po4^{3-}}$ |
| chlorate | $\ce{clo3^-}$ | hydrogen phosphate | $\ce{hpo4^{2-}}$ |
Step1: Identify ions
Ammonium ion: $NH_4^+$ (charge +1). Phosphate ion: $PO_4^{3-}$ (charge -3).
Step2: Balance charges
To balance, find ratio: Let $x$ = number of $NH_4^+$, $y$ = number of $PO_4^{3-}$.
Charge balance: $x(+1) + y(-3) = 0$ → $x = 3y$.
Take $y = 1$, then $x = 3$. So formula: $(NH_4)_3PO_4$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. $(NH_4)_3PO_4$