QUESTION IMAGE
Question
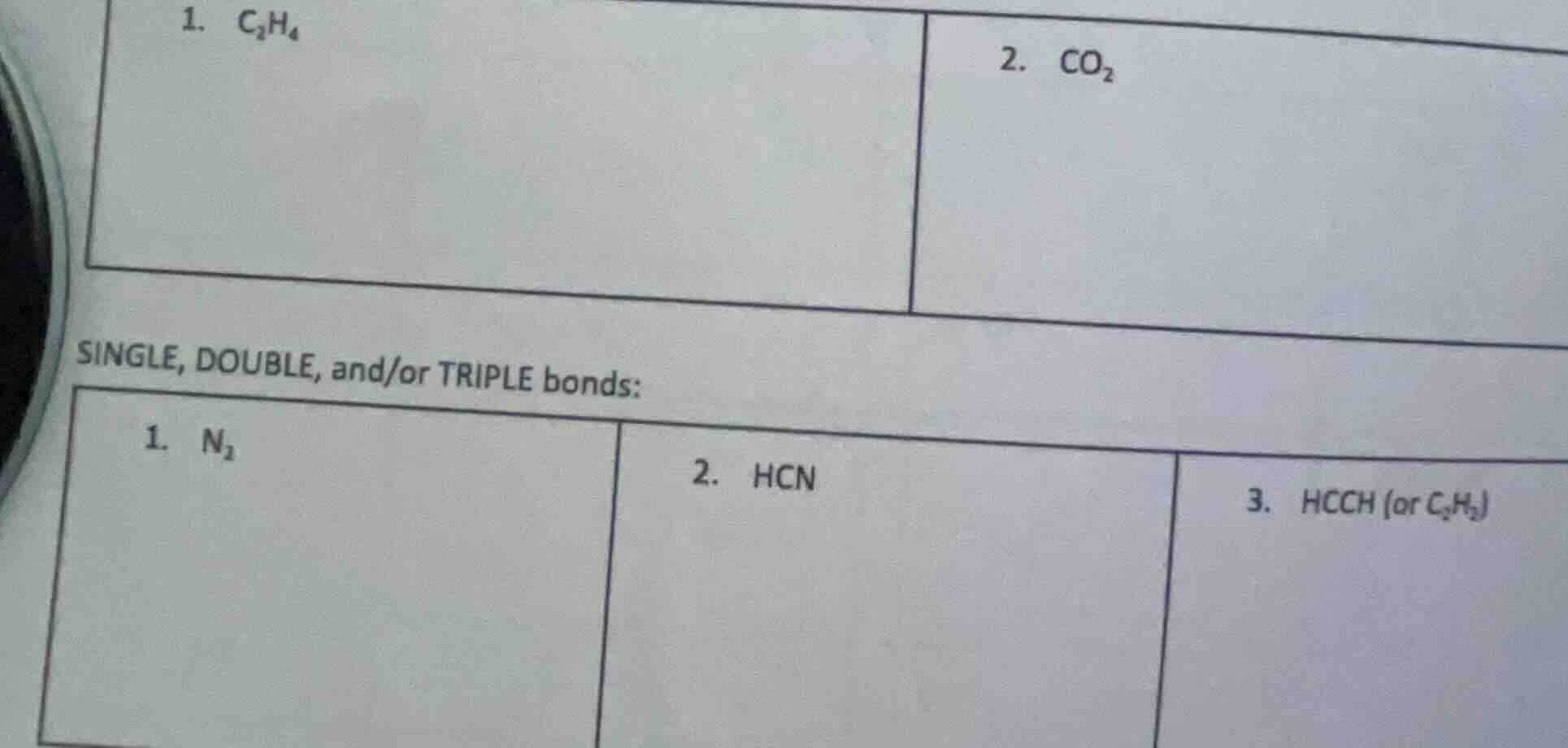
- $c_2h_4$ 2. $co_2$ single, double, and/or triple bonds: 1. $n_2$ 2. $hcn$ 3. $hcch$ (or $c_2h_2$)
Response
To determine the types of bonds (single, double, triple) in each molecule, we analyze their Lewis structures and bonding:
1. $\boldsymbol{C_2H_4}$ (Ethene)
- Step 1: Carbon has 4 valence electrons, hydrogen has 1. For 2 C and 4 H: Total valence electrons = $2(4) + 4(1) = 12$.
- Step 2: The Lewis structure has a double bond between the two C atoms ($\ce{C=C}$) and single bonds between C and H ($\ce{C-H}$).
- Bonds: 1 double bond ($\ce{C=C}$) and 4 single bonds ($\ce{C-H}$).
2. $\boldsymbol{CO_2}$ (Carbon Dioxide)
- Step 1: Carbon has 4, oxygen has 6. Total valence electrons = $4 + 2(6) = 16$.
- Step 2: The Lewis structure has two double bonds: $\ce{O=C=O}$ (each $\ce{C=O}$ is a double bond).
- Bonds: 2 double bonds ($\ce{C=O}$).
3. $\boldsymbol{N_2}$ (Nitrogen Gas)
- Step 1: Each N has 5 valence electrons. Total = $2(5) = 10$.
- Step 2: The Lewis structure has a triple bond: $\ce{N\equiv N}$ (to satisfy octet, 3 shared pairs).
- Bonds: 1 triple bond ($\ce{N\equiv N}$).
4. $\boldsymbol{HCN}$ (Hydrogen Cyanide)
- Step 1: H (1) + C (4) + N (5) = 10 valence electrons.
- Step 2: Lewis structure: $\ce{H-C\equiv N}$ (single bond $\ce{H-C}$, triple bond $\ce{C\equiv N}$).
- Bonds: 1 single bond ($\ce{H-C}$), 1 triple bond ($\ce{C\equiv N}$).
5. $\boldsymbol{HCCH}$ (Ethyne, $\ce{C_2H_2}$)
- Step 1: 2 C (4 each) + 2 H (1 each) = $2(4) + 2(1) = 10$ valence electrons.
- Step 2: Lewis structure: $\ce{H-C\equiv C-H}$ (triple bond between C atoms, single bonds $\ce{C-H}$).
- Bonds: 1 triple bond ($\ce{C\equiv C}$) and 2 single bonds ($\ce{C-H}$).
Summary of Bonds:
| Molecule | Double Bonds | Single Bonds | Triple Bonds |
|---|---|---|---|
| $\ce{CO_2}$ | 2 ($\ce{C=O}$) | 0 | 0 |
| $\ce{N_2}$ | 0 | 0 | 1 ($\ce{N\equiv N}$) |
| $\ce{HCN}$ | 0 | 1 ($\ce{H-C}$) | 1 ($\ce{C\equiv N}$) |
| $\ce{HCCH}$ | 0 | 2 ($\ce{C-H}$) | 1 ($\ce{C\equiv C}$) |
(If you need a specific molecule’s bond type, refer to the table above.)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To determine the types of bonds (single, double, triple) in each molecule, we analyze their Lewis structures and bonding:
1. $\boldsymbol{C_2H_4}$ (Ethene)
- Step 1: Carbon has 4 valence electrons, hydrogen has 1. For 2 C and 4 H: Total valence electrons = $2(4) + 4(1) = 12$.
- Step 2: The Lewis structure has a double bond between the two C atoms ($\ce{C=C}$) and single bonds between C and H ($\ce{C-H}$).
- Bonds: 1 double bond ($\ce{C=C}$) and 4 single bonds ($\ce{C-H}$).
2. $\boldsymbol{CO_2}$ (Carbon Dioxide)
- Step 1: Carbon has 4, oxygen has 6. Total valence electrons = $4 + 2(6) = 16$.
- Step 2: The Lewis structure has two double bonds: $\ce{O=C=O}$ (each $\ce{C=O}$ is a double bond).
- Bonds: 2 double bonds ($\ce{C=O}$).
3. $\boldsymbol{N_2}$ (Nitrogen Gas)
- Step 1: Each N has 5 valence electrons. Total = $2(5) = 10$.
- Step 2: The Lewis structure has a triple bond: $\ce{N\equiv N}$ (to satisfy octet, 3 shared pairs).
- Bonds: 1 triple bond ($\ce{N\equiv N}$).
4. $\boldsymbol{HCN}$ (Hydrogen Cyanide)
- Step 1: H (1) + C (4) + N (5) = 10 valence electrons.
- Step 2: Lewis structure: $\ce{H-C\equiv N}$ (single bond $\ce{H-C}$, triple bond $\ce{C\equiv N}$).
- Bonds: 1 single bond ($\ce{H-C}$), 1 triple bond ($\ce{C\equiv N}$).
5. $\boldsymbol{HCCH}$ (Ethyne, $\ce{C_2H_2}$)
- Step 1: 2 C (4 each) + 2 H (1 each) = $2(4) + 2(1) = 10$ valence electrons.
- Step 2: Lewis structure: $\ce{H-C\equiv C-H}$ (triple bond between C atoms, single bonds $\ce{C-H}$).
- Bonds: 1 triple bond ($\ce{C\equiv C}$) and 2 single bonds ($\ce{C-H}$).
Summary of Bonds:
| Molecule | Double Bonds | Single Bonds | Triple Bonds |
|---|---|---|---|
| $\ce{CO_2}$ | 2 ($\ce{C=O}$) | 0 | 0 |
| $\ce{N_2}$ | 0 | 0 | 1 ($\ce{N\equiv N}$) |
| $\ce{HCN}$ | 0 | 1 ($\ce{H-C}$) | 1 ($\ce{C\equiv N}$) |
| $\ce{HCCH}$ | 0 | 2 ($\ce{C-H}$) | 1 ($\ce{C\equiv C}$) |
(If you need a specific molecule’s bond type, refer to the table above.)