QUESTION IMAGE
Question
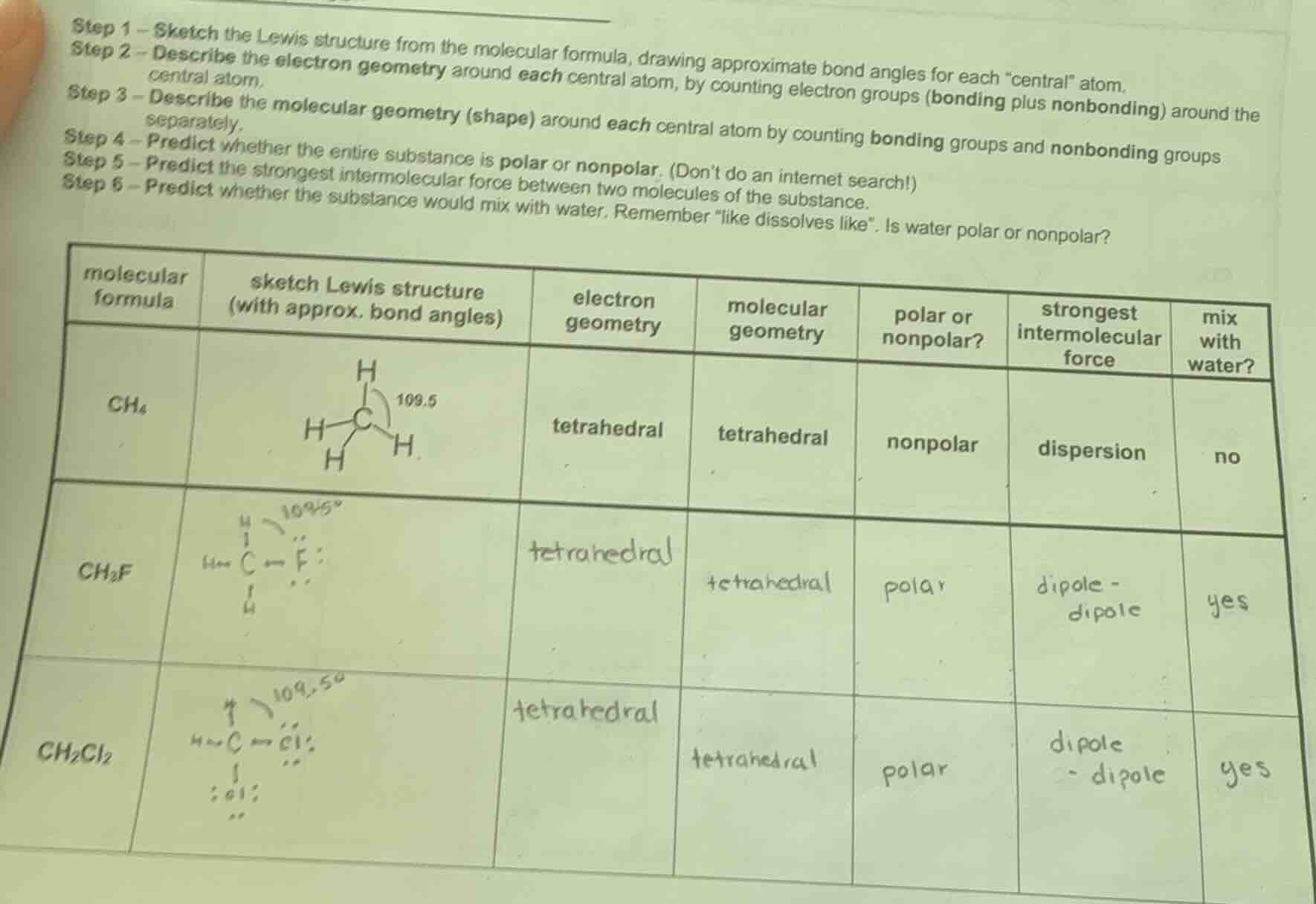
step 1 – sketch the lewis structure from the molecular formula, drawing approximate bond angles for each \central\ atom.
step 2 – describe the electron geometry around each central atom, by counting electron groups (bonding plus nonbonding) around the central atom.
step 3 – describe the molecular geometry (shape) around each central atom by counting bonding groups and nonbonding groups separately.
step 4 – predict whether the entire substance is polar or nonpolar. (dont do an internet search!)
step 5 – predict the strongest intermolecular force between two molecules of the substance.
step 6 – predict whether the substance would mix with water. remember \like dissolves like\. is water polar or nonpolar?
| molecular formula | sketch lewis structure (with approx. bond angles) | electron geometry | molecular geometry | polar or nonpolar? | strongest intermolecular force | mix with water? |
|---|---|---|---|---|---|---|
| $\ce{ch3f}$ | <img src=\https://via.placeholder.com/100x50?text=109.5%c2%b0+c-f\ /> | tetrahedral | tetrahedral | polar | dipole - dipole | yes |
| $\ce{ch2cl2}$ | <img src=\https://via.placeholder.com/100x50?text=109.5%c2%b0+c-cl\ /> | tetrahedral | tetrahedral | polar | dipole - dipole | yes |
Step1: Verify CH₄ entries
Lewis structure: $\ce{H-\underset{\underset{H}{|}}{C}-H}$ (bond angle $109.5^\circ$), electron groups = 4 bonding, 0 lone pairs.
Electron geometry: tetrahedral, molecular geometry: tetrahedral. Symmetric, so nonpolar. Only dispersion forces. Nonpolar does not mix with polar water.
Step2: Verify CH₃F entries
Lewis structure: $\ce{H-\underset{\underset{H}{|}}{C}-F:}$ (bond angle $109.5^\circ$), electron groups = 4 bonding, 0 lone pairs.
Electron geometry: tetrahedral, molecular geometry: tetrahedral. Unequal electronegativity creates dipole, polar. Dipole-dipole forces. Polar mixes with polar water.
Step3: Verify CH₂Cl₂ entries
Lewis structure: $\ce{:\!Cl-\underset{\underset{H}{|}}{C}-Cl:}$ (bond angle $109.5^\circ$), electron groups = 4 bonding, 0 lone pairs.
Electron geometry: tetrahedral, molecular geometry: tetrahedral. Unequal electronegativity creates net dipole, polar. Dipole-dipole forces. Polar mixes with polar water.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
All entries in the table are verified as correct:
- For $\ce{CH4}$: Lewis structure (109.5° bond angle), electron geometry = tetrahedral, molecular geometry = tetrahedral, nonpolar, strongest intermolecular force = dispersion, does not mix with water.
- For $\ce{CH3F}$: Lewis structure (109.5° bond angle), electron geometry = tetrahedral, molecular geometry = tetrahedral, polar, strongest intermolecular force = dipole-dipole, mixes with water.
- For $\ce{CH2Cl2}$: Lewis structure (109.5° bond angle), electron geometry = tetrahedral, molecular geometry = tetrahedral, polar, strongest intermolecular force = dipole-dipole, mixes with water.