QUESTION IMAGE
Question
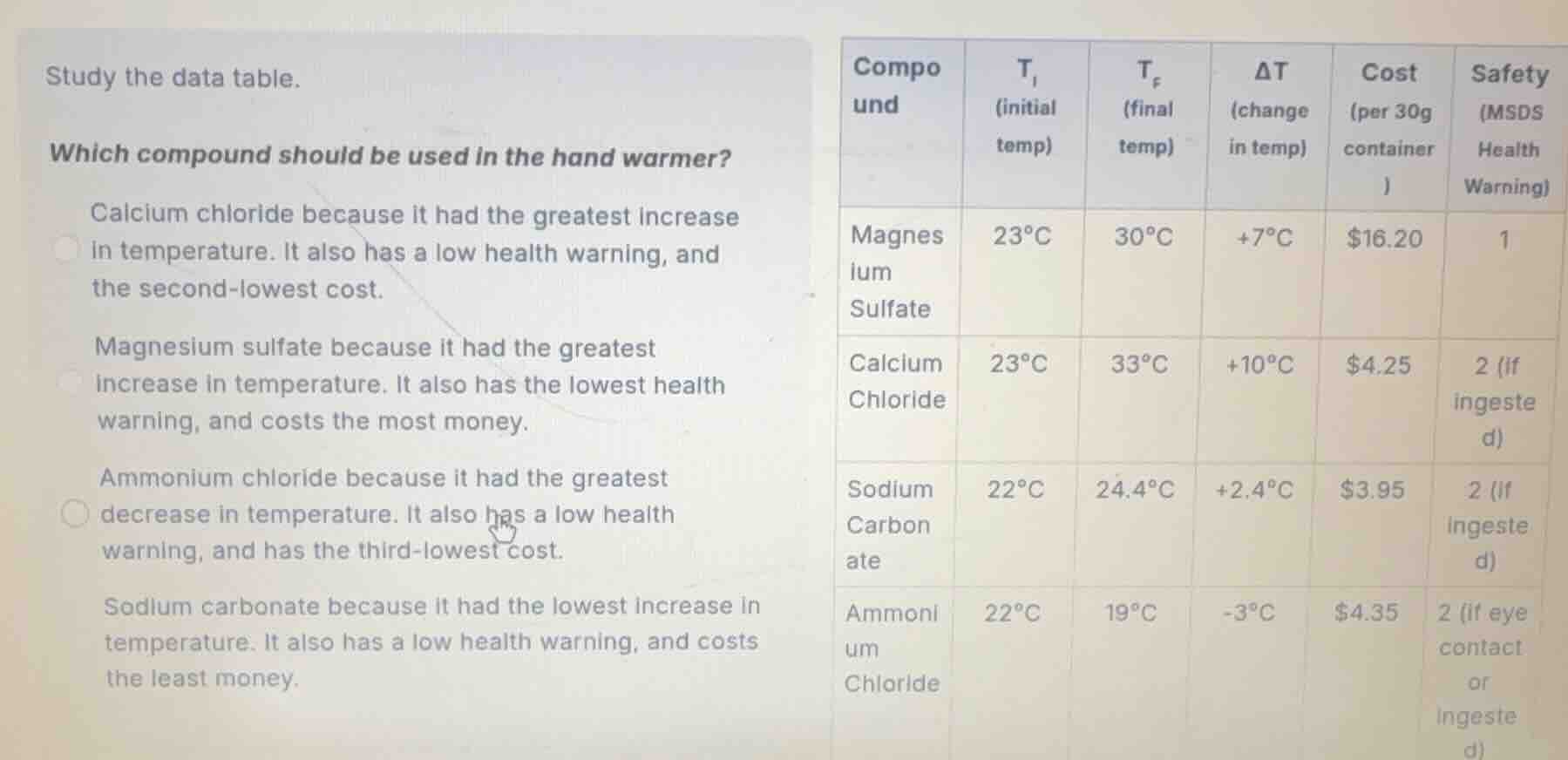
study the data table.
which compound should be used in the hand warmer?
calcium chloride because it had the greatest increase in temperature. it also has a low health warning, and the second-lowest cost.
magnesium sulfate because it had the greatest increase in temperature. it also has the lowest health warning, and costs the most money.
ammonium chloride because it had the greatest decrease in temperature. it also has a low health warning, and has the third-lowest cost.
sodium carbonate because it had the lowest increase in temperature. it also has a low health warning, and costs the least money.
| compound | $t_i$ (initial temp) | $t_f$ (final temp) | $delta t$ (change in temp) | cost (per 30g container) | safety (msds health warning) | ---- | ---- | ---- | ---- | ---- | ---- | magnesium sulfate | $23^circ c$ | $30^circ c$ | $+7^circ c$ | $16.20$ | 1 | calcium chloride | $23^circ c$ | $33^circ c$ | $+10^circ c$ | $4.25$ | 2 (if ingested) | sodium carbonate | $22^circ c$ | $24.4^circ c$ | $+2.4^circ c$ | $3.95$ | 2 (if ingested) | ammonium chloride | $22^circ c$ | $19^circ c$ | $-3^circ c$ | $4.35$ | 2 (if eye contact or ingested) |
A hand warmer needs a compound that releases heat, shown by a positive temperature increase ($\Delta T$). First, eliminate ammonium chloride (it cools, $\Delta T=-3^\circ\text{C}$). Next, compare the remaining compounds:
- Calcium chloride has the largest temperature increase ($+10^\circ\text{C}$), meaning it releases the most heat.
- Its cost ($\$4.25$) is the second-lowest, making it affordable.
- Its health warning is only a risk if ingested, which is manageable with proper use.
The other options are incorrect: magnesium sulfate has a smaller temperature rise, sodium carbonate has a very small heat release, and ammonium chloride cools instead of warming.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Calcium chloride because it had the greatest increase in temperature. It also has a low health warning, and the second-lowest cost.