QUESTION IMAGE
Question
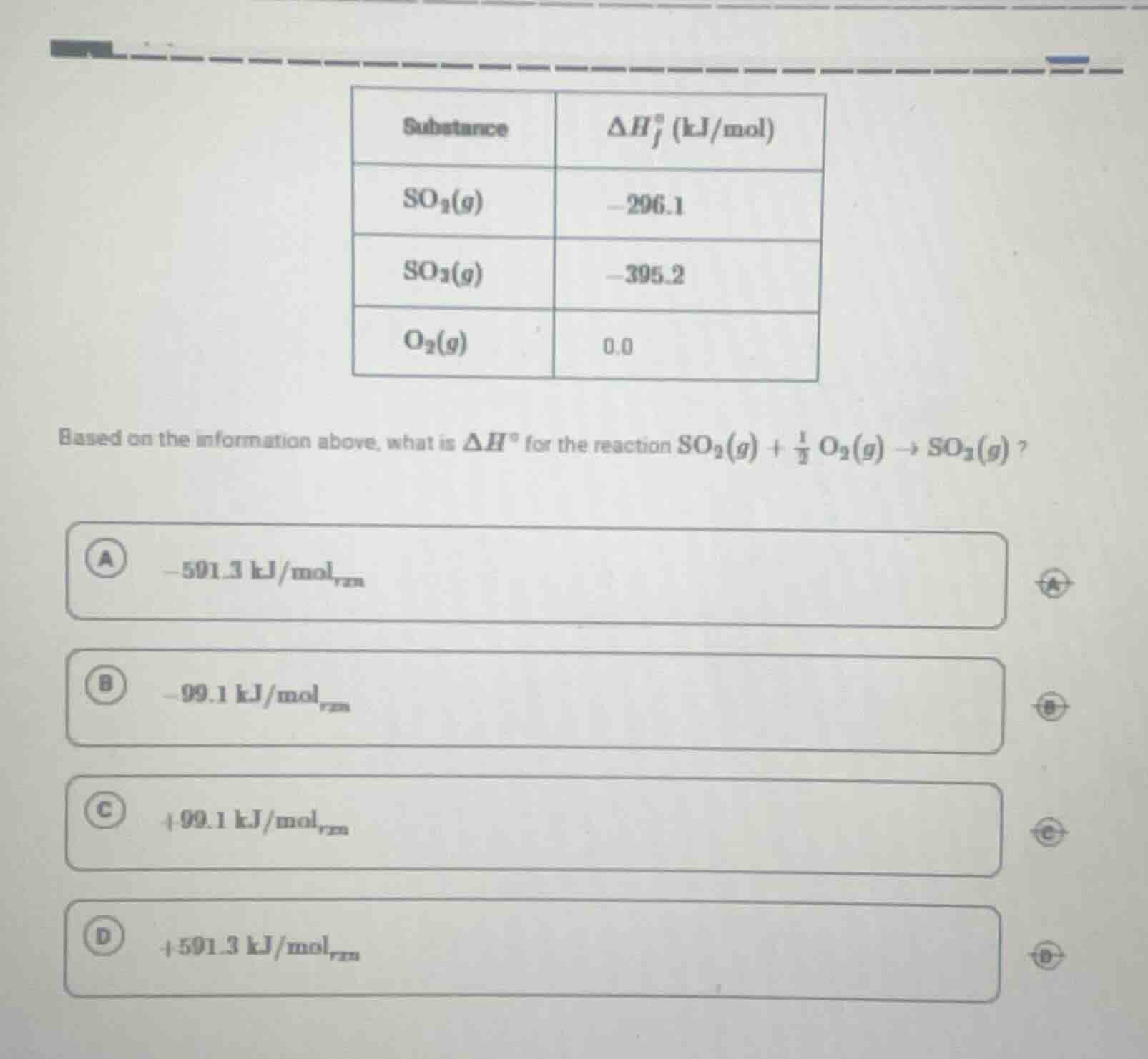
substance | $delta h_f^circ$ (kj/mol)
--- | ---
$so_2(g)$ | -296.1
$so_3(g)$ | -395.2
$o_2(g)$ | 0.0
based on the information above, what is $delta h^circ$ for the reaction $so_2(g) + \frac{1}{2} o_2(g) \
ightarrow so_3(g)$?
a) -591.3 kj/mol$_{rxn}$
b) -99.1 kj/mol$_{rxn}$
c) +99.1 kj/mol$_{rxn}$
d) +591.3 kj/mol$_{rxn}$
Step1: Recall the formula for enthalpy of reaction
The enthalpy of a reaction ($\Delta H^{\circ}$) is calculated as the sum of the enthalpies of formation of the products minus the sum of the enthalpies of formation of the reactants. The formula is $\Delta H^{\circ} = \sum \Delta H_f^{\circ}(\text{products}) - \sum \Delta H_f^{\circ}(\text{reactants})$.
Step2: Identify reactants and products
For the reaction $\text{SO}_2(g) + \frac{1}{2}\text{O}_2(g)
ightarrow \text{SO}_3(g)$, the product is $\text{SO}_3(g)$ and the reactants are $\text{SO}_2(g)$ and $\text{O}_2(g)$.
Step3: Substitute values into the formula
From the table, $\Delta H_f^{\circ}(\text{SO}_3(g)) = -395.2\ \text{kJ/mol}$, $\Delta H_f^{\circ}(\text{SO}_2(g)) = -296.1\ \text{kJ/mol}$, and $\Delta H_f^{\circ}(\text{O}_2(g)) = 0.0\ \text{kJ/mol}$ (since elemental oxygen in its standard state has $\Delta H_f^{\circ} = 0$).
So, $\sum \Delta H_f^{\circ}(\text{products}) = \Delta H_f^{\circ}(\text{SO}_3(g)) = -395.2\ \text{kJ/mol}$.
$\sum \Delta H_f^{\circ}(\text{reactants}) = \Delta H_f^{\circ}(\text{SO}_2(g)) + \frac{1}{2}\Delta H_f^{\circ}(\text{O}_2(g)) = -296.1 + \frac{1}{2}(0.0) = -296.1\ \text{kJ/mol}$.
Now, $\Delta H^{\circ} = (-395.2) - (-296.1) = -395.2 + 296.1 = -99.1\ \text{kJ/mol}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. $-99.1\ \text{kJ/mol}_{\text{rxn}}$