QUESTION IMAGE
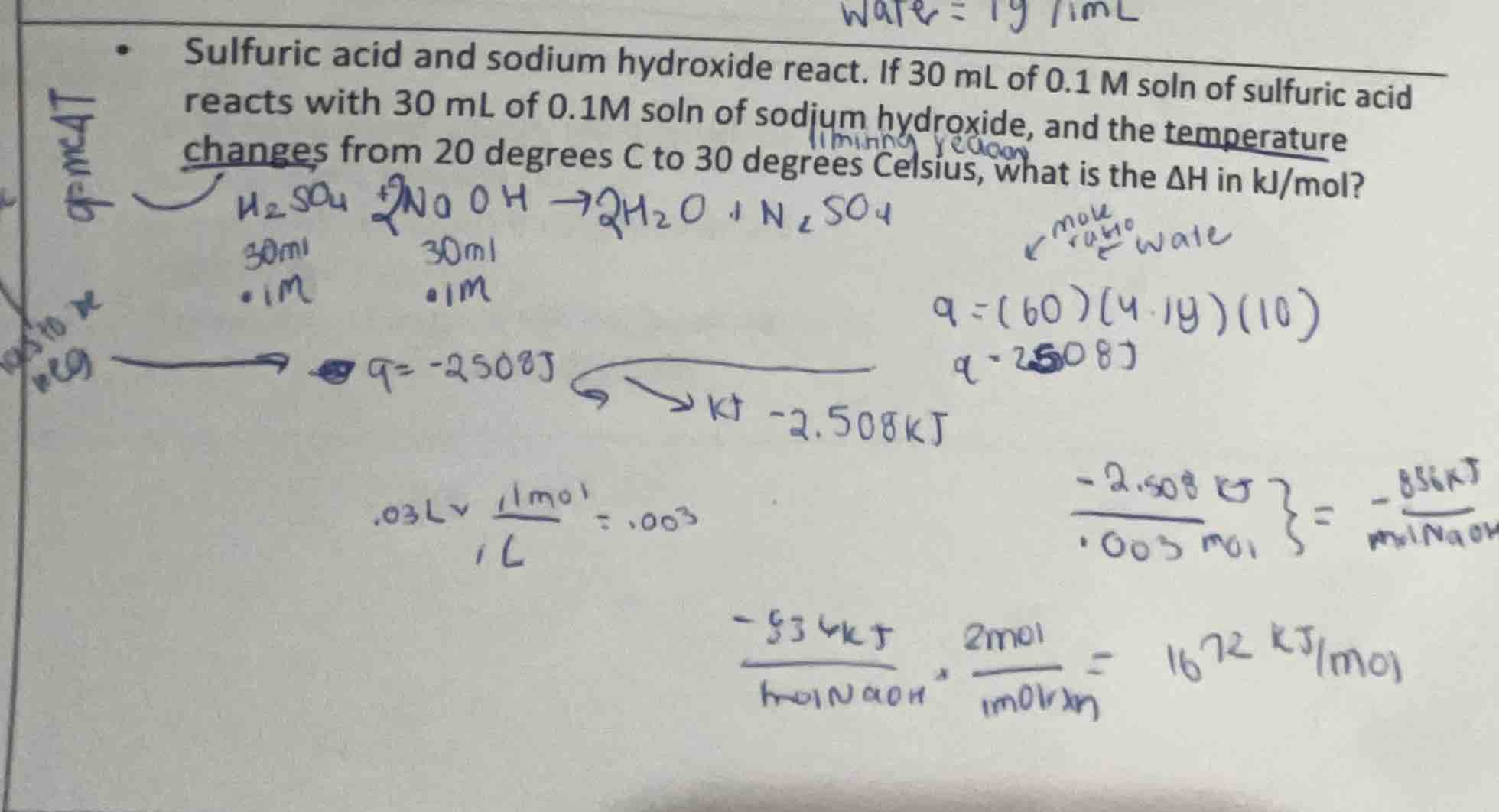
Question
sulfuric acid and sodium hydroxide react. if 30 ml of 0.1 m soln of sulfuric acid reacts with 30 ml of 0.1m soln of sodium hydroxide, and the temperature changes from 20 degrees c to 30 degrees celsius, what is the δh in kj/mol?
Step1: Calculate total volume of solution
The volume of sulfuric acid is \( 30\space mL \) and sodium hydroxide is \( 30\space mL \), so total volume \( V = 30 + 30=60\space mL \). Since density of water (solution) is \( 1\space g/mL \), mass \( m = 60\space g \) (as \( mass = density\times volume \), \( density = 1\space g/mL \), \( volume = 60\space mL \), so \( m = 60\times1 = 60\space g \)).
Step2: Calculate heat absorbed by solution (\( q_{solution} \))
Using formula \( q = mc\Delta T \), where \( c = 4.18\space J/g^\circ C \) (specific heat of water), \( \Delta T=30 - 20 = 10^\circ C \), \( m = 60\space g \).
\( q_{solution}=60\times4.18\times10 = 2508\space J=2.508\space kJ \)
Step3: Determine heat released by reaction (\( q_{reaction} \))
The reaction is exothermic, so \( q_{reaction}=-q_{solution}=- 2.508\space kJ \)
Step4: Calculate moles of reactants
Moles of \( H_2SO_4 = M\times V(L)=0.1\times0.03 = 0.003\space mol \)
Moles of \( NaOH = 0.1\times0.03 = 0.003\space mol \)
The reaction is \( H_2SO_4 + 2NaOH
ightarrow Na_2SO_4 + 2H_2O \). From stoichiometry, 1 mol \( H_2SO_4 \) reacts with 2 mol \( NaOH \). Here, \( NaOH \) is the limiting reactant (0.003 mol \( NaOH \) will react with \( \frac{0.003}{2}=0.0015\space mol \) of \( H_2SO_4 \))
Step5: Calculate \( \Delta H \) per mole of reaction
Moles of reaction (based on \( NaOH \)): For 2 mol \( NaOH \), 1 mol reaction occurs. So for 0.003 mol \( NaOH \), moles of reaction \( n=\frac{0.003}{2}=0.0015\space mol \)
\( \Delta H=\frac{q_{reaction}}{n}=\frac{- 2.508}{0.0015}=- 1672\space kJ/mol \) (the negative sign indicates exothermic reaction)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(\Delta H=- 1672\space kJ/mol\) (or \(1672\space kJ/mol\) released, so \(\Delta H = - 1672\space kJ/mol\))