QUESTION IMAGE
Question
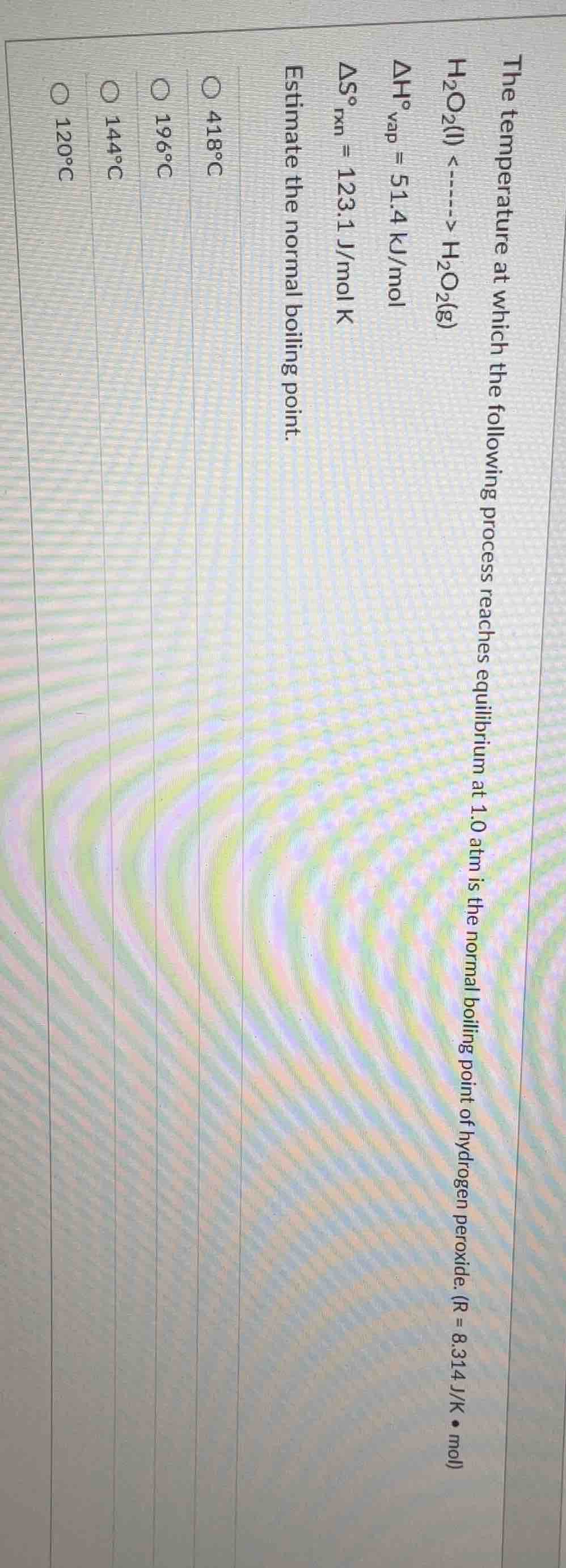
the temperature at which the following process reaches equilibrium at 1.0 atm is the normal boiling point of hydrogen peroxide. (r = 8.314 j/k • mol)
h₂o₂(l) <-----> h₂o₂(g)
δh°vap = 51.4 kj/mol
δs°rxn = 123.1 j/mol k
estimate the normal boiling point.
○ 418°c
○ 196°c
○ 144°c
○ 120°c
Step1: Recall the equilibrium condition for phase change
At normal boiling point (equilibrium at 1.0 atm), the Gibbs free energy change \(\Delta G^{\circ} = 0\). The relationship between \(\Delta G^{\circ}\), \(\Delta H^{\circ}\), and \(\Delta S^{\circ}\) is \(\Delta G^{\circ}=\Delta H^{\circ}-T\Delta S^{\circ}\). Setting \(\Delta G^{\circ} = 0\), we get \(0=\Delta H^{\circ}-T\Delta S^{\circ}\), which can be rearranged to \(T=\frac{\Delta H^{\circ}}{\Delta S^{\circ}}\).
Step2: Convert units of \(\Delta H^{\circ}\)
Given \(\Delta H^{\circ}_{\text{vap}} = 51.4\space kJ/mol\). To use the same units as \(\Delta S^{\circ}\) (which is in \(J/mol\space K\)), convert \(kJ\) to \(J\): \(51.4\space kJ/mol=51.4\times1000\space J/mol = 51400\space J/mol\).
Step3: Calculate the temperature in Kelvin
Substitute \(\Delta H^{\circ}=51400\space J/mol\) and \(\Delta S^{\circ}=123.1\space J/mol\space K\) into the formula \(T=\frac{\Delta H^{\circ}}{\Delta S^{\circ}}\):
\[T=\frac{51400\space J/mol}{123.1\space J/mol\space K}\approx417.55\space K\]
Step4: Convert temperature from Kelvin to Celsius
Use the conversion formula \(T(^{\circ}C)=T(K)-273.15\). So, \(T(^{\circ}C)=417.55 - 273.15\approx144.4^{\circ}C\approx144^{\circ}C\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
144°C (corresponding to the option: 144°C)