QUESTION IMAGE
Question
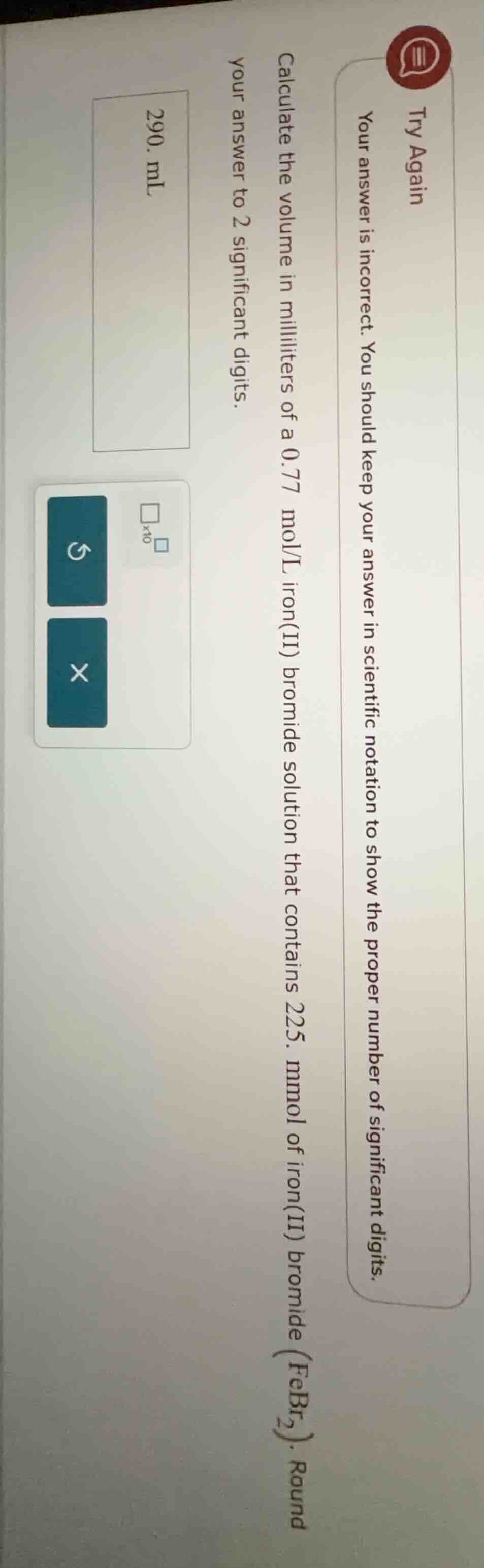
try again
your answer is incorrect. you should keep your answer in scientific notation to show the proper number of significant digits.
calculate the volume in milliliters of a 0.77 mol/l iron(ii) bromide solution that contains 225. mmol of iron(ii) bromide ($febr_2$). round your answer to 2 significant digits.
- ml
Step1: Convert mmol to mol
$225. \text{ mmol} = 225. \times 10^{-3} \text{ mol} = 0.225 \text{ mol}$
Step2: Use molarity formula to find volume
Molarity $M = \frac{n}{V}$, rearrange to $V = \frac{n}{M}$
$V = \frac{0.225 \text{ mol}}{0.77 \text{ mol/L}} \approx 0.2922 \text{ L}$
Step3: Convert liters to milliliters
$0.2922 \text{ L} \times 1000 = 292.2 \text{ mL}$
Step4: Round to 2 sig figs, write in scientific notation
$292.2 \text{ mL} \approx 2.9 \times 10^2 \text{ mL}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$2.9 \times 10^2$ mL