QUESTION IMAGE
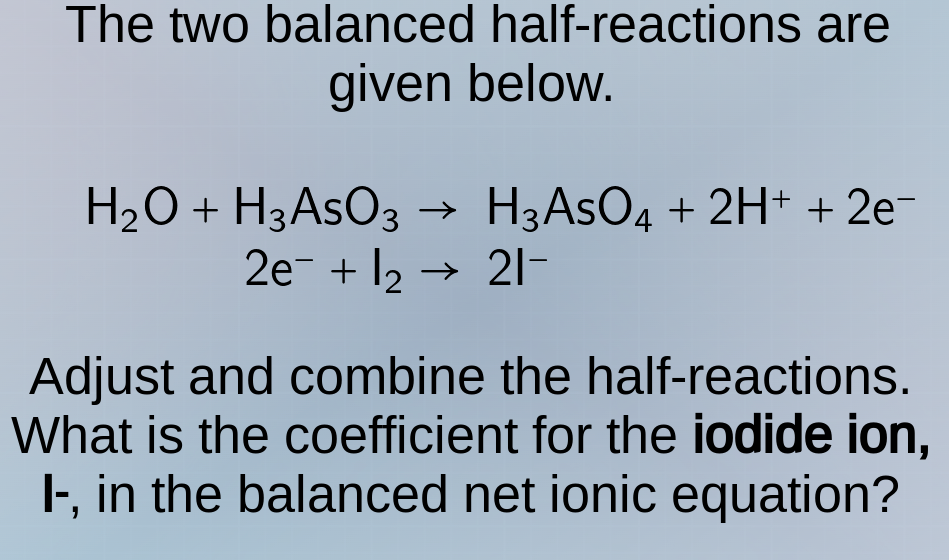
Question
the two balanced half - reactions are given below.
$ce{h_{2}o + h_{3}aso_{3} -> h_{3}aso_{4} + 2h^{+} + 2e^{-}}$
$ce{2e^{-} + i_{2} -> 2i^{-}}$
adjust and combine the half - reactions. what is the coefficient for the iodide ion, $ce{i^{-}}$, in the balanced net ionic equation?
Step1: Check electron transfer
The first half - reaction (oxidation: \(H_2O + H_3AsO_3
ightarrow H_3AsO_4+2H^++2e^-\)) releases 2 electrons, and the second half - reaction (reduction: \(2e^- + I_2
ightarrow 2I^-\)) gains 2 electrons. The number of electrons lost in oxidation is equal to the number of electrons gained in reduction.
Step2: Combine the half - reactions
To get the net ionic equation, we add the two half - reactions together. When we add \(H_2O + H_3AsO_3
ightarrow H_3AsO_4+2H^++2e^-\) and \(2e^- + I_2
ightarrow 2I^-\), the electrons (\(2e^-\)) cancel out. So the combined equation is \(H_2O + H_3AsO_3+I_2
ightarrow H_3AsO_4 + 2H^++2I^-\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2