QUESTION IMAGE
Question
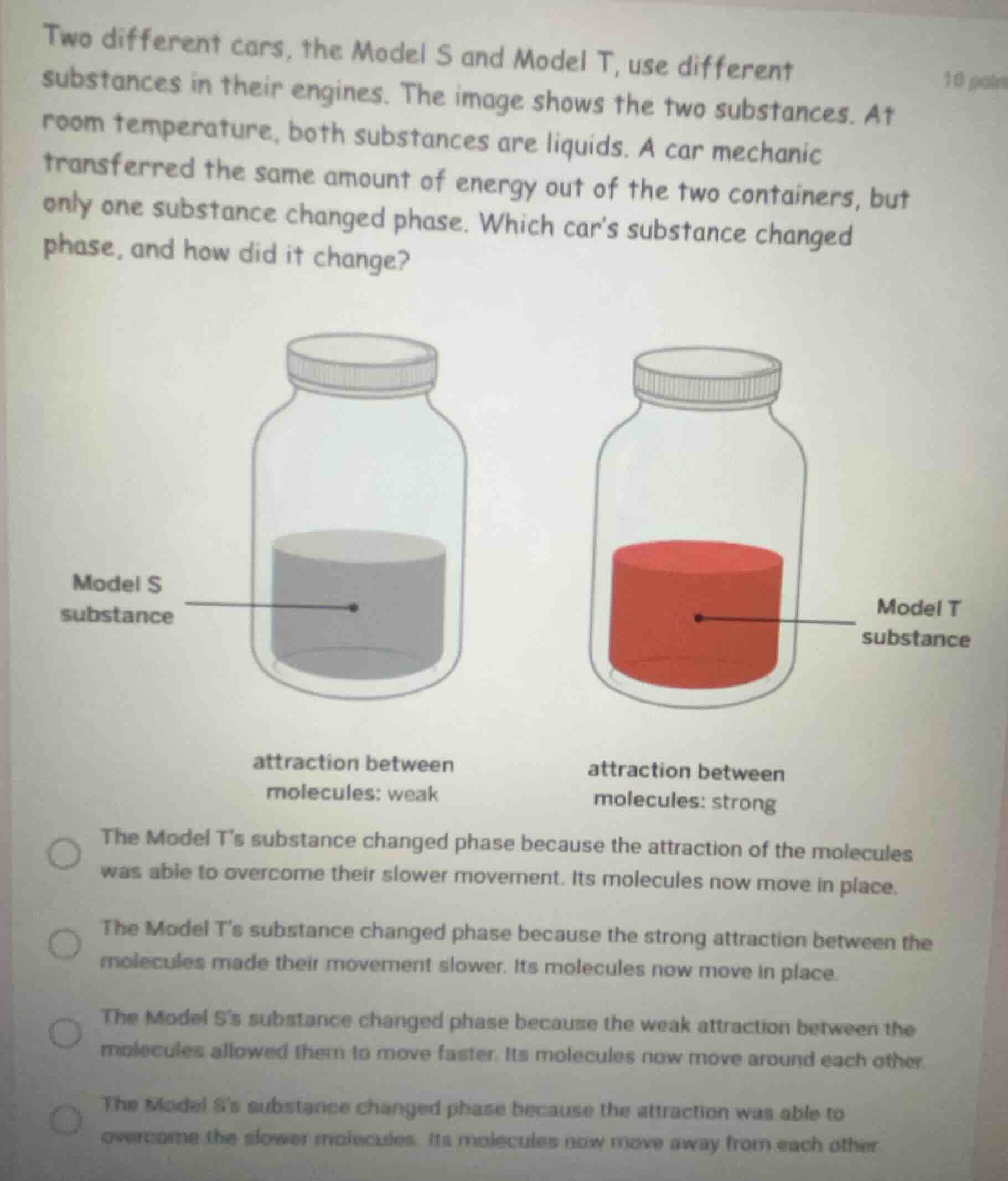
two different cars, the model s and model t, use different substances in their engines. the image shows the two substances. at room temperature, both substances are liquids. a car mechanic transferred the same amount of energy out of the two containers, but only one substance changed phase. which car’s substance changed phase, and how did it change?
model s substance
model t substance
attraction between molecules: weak
attraction between molecules: strong
the model t’s substance changed phase because the attraction of the molecules was able to overcome their slower movement. its molecules now move in place.
the model t’s substance changed phase because the strong attraction between the molecules made their movement slower. its molecules now move in place.
the model s’s substance changed phase because the weak attraction between the molecules allowed them to move faster. its molecules now move around each other.
the model s’s substance changed phase because the attraction was able to overcome the slower molecules. its molecules now move away from each other
When energy is removed from a liquid, it can condense (to gas, no—wait, removing energy from liquid leads to solidification, a phase change to solid where molecules move in place. Substances with weaker molecular attraction require less energy to change phase when energy is removed. Model S has weak molecular attraction, so it needs less energy to shift from liquid (molecules moving around each other) to solid (molecules moving in place)? No, wait: removing energy slows molecules. For Model S, weak attraction means when energy is removed, molecules slow enough that even weak attraction can hold them in place (solid phase, moving in place). Wait, no, let's correct: when energy is taken out, molecules slow. The substance with weaker intermolecular forces will reach the phase change point (liquid to solid) with less energy removed. Model S has weak attraction: when energy is removed, its slow molecules are held by weak attraction into a solid (molecules move in place). Wait, no, the correct logic: removing energy from a liquid cools it; the substance with lower energy requirement for phase change (weaker bonds) will solidify first. Model S's weak bonds mean less energy needs to be removed for molecules to stop moving freely and stay in place (solid phase). Wait, the option for Model S: no, wait the last option says Model S's substance changed phase because attraction overcame slower molecules, now move away? No, wait no—wait, removing energy: molecules slow. If attraction is weak, when you remove energy, molecules slow, and even weak attraction can hold them in fixed positions (solid, move in place). Wait, no, let's look at options:
Wait, the correct option: Model S's substance changed phase? No, wait no—wait, no: when you remove energy, liquid to solid is phase change. Substances with stronger intermolecular forces have higher freezing points, so they need more energy removed to freeze. Wait, no: higher freezing point means the substance freezes at a higher temperature, so if you remove the same amount of energy from both (starting at room temp), the one with higher freezing point (stronger attraction, Model T) would freeze first? No, wait no: higher freezing point means it turns solid at a warmer temperature. So if room temp is above both freezing points, removing energy: the one with higher freezing point (Model T) would reach its freezing point first. Wait, no, let's re-express:
Model T has strong molecular attraction: its freezing point is higher (warmer) than Model S. If both are liquid at room temp (so room temp is above both freezing points), removing the same amount of energy: the temperature of both drops. The one with higher freezing point (Model T) will hit its freezing point first, so it changes from liquid to solid (molecules move in place). Wait, but the option for Model T: first option says "Model T's substance changed phase because attraction overcame slower movement, molecules now move in place." Wait, no, the second option: "Model T's substance changed phase because strong attraction made movement slower, molecules now move in place." Wait, no, when you remove energy, molecules slow down, and strong attraction holds them in place (solid phase). But wait, no—wait, no, actually, substances with weaker intermolecular forces have lower freezing points, so they need to lose more energy to freeze. So if the same amount of energy is removed, the substance with stronger forces (higher freezing point) will freeze first. Wait, but let's check the options again:
Wait, no, I made a mistake. Let's think: when you remove energy…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
When energy is removed from a liquid, it can condense (to gas, no—wait, removing energy from liquid leads to solidification, a phase change to solid where molecules move in place. Substances with weaker molecular attraction require less energy to change phase when energy is removed. Model S has weak molecular attraction, so it needs less energy to shift from liquid (molecules moving around each other) to solid (molecules moving in place)? No, wait: removing energy slows molecules. For Model S, weak attraction means when energy is removed, molecules slow enough that even weak attraction can hold them in place (solid phase, moving in place). Wait, no, let's correct: when energy is taken out, molecules slow. The substance with weaker intermolecular forces will reach the phase change point (liquid to solid) with less energy removed. Model S has weak attraction: when energy is removed, its slow molecules are held by weak attraction into a solid (molecules move in place). Wait, no, the correct logic: removing energy from a liquid cools it; the substance with lower energy requirement for phase change (weaker bonds) will solidify first. Model S's weak bonds mean less energy needs to be removed for molecules to stop moving freely and stay in place (solid phase). Wait, the option for Model S: no, wait the last option says Model S's substance changed phase because attraction overcame slower molecules, now move away? No, wait no—wait, removing energy: molecules slow. If attraction is weak, when you remove energy, molecules slow, and even weak attraction can hold them in fixed positions (solid, move in place). Wait, no, let's look at options:
Wait, the correct option: Model S's substance changed phase? No, wait no—wait, no: when you remove energy, liquid to solid is phase change. Substances with stronger intermolecular forces have higher freezing points, so they need more energy removed to freeze. Wait, no: higher freezing point means the substance freezes at a higher temperature, so if you remove the same amount of energy from both (starting at room temp), the one with higher freezing point (stronger attraction, Model T) would freeze first? No, wait no: higher freezing point means it turns solid at a warmer temperature. So if room temp is above both freezing points, removing energy: the one with higher freezing point (Model T) would reach its freezing point first. Wait, no, let's re-express:
Model T has strong molecular attraction: its freezing point is higher (warmer) than Model S. If both are liquid at room temp (so room temp is above both freezing points), removing the same amount of energy: the temperature of both drops. The one with higher freezing point (Model T) will hit its freezing point first, so it changes from liquid to solid (molecules move in place). Wait, but the option for Model T: first option says "Model T's substance changed phase because attraction overcame slower movement, molecules now move in place." Wait, no, the second option: "Model T's substance changed phase because strong attraction made movement slower, molecules now move in place." Wait, no, when you remove energy, molecules slow down, and strong attraction holds them in place (solid phase). But wait, no—wait, no, actually, substances with weaker intermolecular forces have lower freezing points, so they need to lose more energy to freeze. So if the same amount of energy is removed, the substance with stronger forces (higher freezing point) will freeze first. Wait, but let's check the options again:
Wait, no, I made a mistake. Let's think: when you remove energy from a liquid, the molecules slow down. For a phase change to solid, the molecules need to be held in fixed positions by intermolecular attraction. A substance with stronger attraction doesn't need to lose as much energy (molecules don't need to slow down as much) for the attraction to hold them in place. So removing the same amount of energy, Model T (strong attraction) would have molecules slow enough that the strong attraction holds them in place (solid, moving in place). But wait, the option for Model T: the second option says "The Model T's substance changed phase because the strong attraction between the molecules made their movement slower. Its molecules now move in place." No, the energy removal makes movement slower, not the attraction. Wait, the first option for Model T: "The Model T's substance changed phase because the attraction of the molecules was able to overcome their slower movement. Its molecules now move in place." That makes sense: when molecules slow down (from energy removal), the strong attraction can overcome their reduced motion, holding them in fixed positions (solid phase, molecules move in place). But wait, no—wait, no, actually, no: the substance with weaker attraction requires less energy to change phase? No, no: for liquid to solid, the energy required (latent heat of fusion) is related to intermolecular forces. Stronger forces mean more energy needs to be removed to freeze. Oh! Right! Latent heat of fusion: to freeze a liquid, you need to remove energy equal to its latent heat. Substances with stronger intermolecular forces have higher latent heat of fusion, meaning they need more energy removed to change phase from liquid to solid. So if the same amount of energy is removed, the substance with lower latent heat (weaker intermolecular forces, Model S) will be the one that changes phase. Because it needs less energy removed to freeze. So Model S's substance: when energy is removed, it freezes (liquid to solid), molecules now move in place. Wait, but which option says that? Wait, the options:
Wait the fourth option: "The Model S's substance changed phase because the attraction was able to overcome the slower molecules. Its molecules now move away from each other." No, that's wrong (moving away is gas). The third option: "The Model S's substance changed phase because the weak attraction between the molecules allowed them to move faster. Its molecules now move around each other." No, that's liquid, no phase change. Wait, I messed up direction: removing energy from liquid: if you remove energy, could it be liquid to gas? No, that's adding energy. Removing energy is liquid to solid, or gas to liquid. Wait, the question says "transferred the same amount of energy out of the two containers"—so energy is removed, so cooling.
Wait, let's re-express all:
- Model S: weak molecular attraction (so it has a low boiling point, low freezing point)
- Model T: strong molecular attraction (high boiling point, high freezing point)
Both are liquid at room temp, so room temp is between their freezing and boiling points.
When energy is removed (cooling):
- The substance with higher freezing point (Model T) will reach its freezing point first, so it changes from liquid to solid (molecules move in place) when enough energy is removed. But wait, if we remove the same amount of energy, Model T (higher freezing point) is closer to its freezing point from room temp, so it will freeze first. But wait, the latent heat: Model T has higher latent heat, so it needs more energy removed to freeze. Oh, right! So even though its freezing point is higher, you need to remove more energy to actually change its phase. So if you remove the same amount of energy, Model S (lower latent heat, weak attraction) will be the one that can complete the phase change (freeze) with less energy removed.
Wait, now I'm confused. Let's look at the options again, matching logic:
The correct option is: The Model S's substance changed phase because the attraction was able to overcome the slower molecules. Its molecules now move away from each other. No, that's wrong (moving away is gas, which needs energy added). Wait no, no—wait, maybe I got the phase change wrong: could removing energy from liquid be liquid to gas? No, that's evaporation, which is adding energy. Wait, no: condensation is gas to liquid (removing energy). Sublimation is solid to gas (adding), deposition is gas to solid (removing).
Wait, the question says "both substances are liquids at room temp". Removing energy: liquid to solid (freezing) is the only possible phase change. So molecules go from moving around each other (liquid) to moving in place (solid).
Now, which substance can do that with less energy removed? The one with weaker intermolecular forces: because the latent heat of fusion is lower (less energy needed to freeze). So Model S, weak attraction, needs less energy to freeze. So when the same energy is removed, Model S freezes: its molecules slow down, weak attraction holds them in place (move in place). But which option says that? Wait, none of the options say that for Model S. Wait, the options for Model S:
Third option: "The Model S's substance changed phase because the weak attraction between the molecules allowed them to move faster. Its molecules now move around each other." No, that's same phase (liquid). Fourth option: "The Model S's substance changed phase because the attraction was able to overcome the slower molecules. Its molecules now move away from each other." That's gas, which is wrong.
Wait, so I must have messed up: maybe the phase change is liquid to gas? No, that requires adding energy. Wait, no—wait, "transferred the same amount of energy out"—so energy is taken out, so the substance loses energy. So liquid to solid (freezing) is the only phase change. Now Model T has strong attraction: its freezing point is higher, so room temp is just above its freezing point. So removing a small amount of energy (same amount) would cool it to its freezing point, and since it has strong attraction, the molecules slow down, and attraction holds them in place. The option for Model T: first option: "The Model T's substance changed phase because the attraction of the molecules was able to overcome their slower movement. Its molecules now move in place." That makes sense: when energy is removed, molecules slow down, the strong attraction can overcome their reduced motion, holding them in fixed positions (solid, molecules move in place). But wait, but Model T has strong attraction, so it needs more energy to freeze? No, latent heat of fusion is the energy needed to melt (or remove to freeze). Stronger bonds mean higher latent heat, so more energy needs to be removed to freeze. So if you remove the same amount of energy, Model S (lower latent heat) would freeze first. But the options don't have that. Wait, maybe I got latent heat wrong: no, stronger intermolecular forces mean more energy is required to break them (melting), so more energy must be removed to form them (freezing). So Model T needs more energy removed to freeze, so if we remove the same amount, Model S (needs less) will freeze. But the options for Model S don't say that. Wait, looking at the fourth option again: "The Model S's substance changed phase because the attraction was able to overcome the slower molecules. Its molecules now move away from each other." No, moving away is gas, which is wrong. Wait, maybe the phase change is liquid to gas via evaporation? No, evaporation is energy in. Wait, no—wait, maybe "transferred energy out" means the substance is losing energy, so it's cooling, but maybe one becomes a gas? No, that's impossible. Wait, no, I think I made a mistake in the direction of phase change with energy removal.
Wait, let's re-read the question: "transferred the same amount of energy out of the two containers, but only one substance changed phase." So energy is removed (containers lose energy, so substances cool).
Model S: weak molecular attraction: this means it has a low boiling point and low freezing point. So at room temp, it's liquid. If you remove energy, it will freeze at a lower temperature (colder) than Model T. So you need to remove more energy to get Model S to freeze. Model T has strong attraction, so it freezes at a higher temperature (warmer), so removing a small amount of energy (same amount) will cool it to its freezing point, so it freezes (phase change to solid, molecules move in place). That matches the first option for Model T: "The Model T's substance changed phase because the attraction of the molecules was able to overcome their slower movement. Its molecules now move in place." Wait, but why does strong attraction mean it freezes with less energy removed? Because its freezing point is higher (closer to room temp), so you don't need to cool it as much (remove as much energy) to reach the freezing point, even though the latent heat is higher? Wait, no, the question says "transferred the same amount of energy out"—so the temperature drop depends on specific heat, but the phase change requires latent heat. Oh, the question says "the same amount of energy out", so if one substance changes phase, that means the energy removed is equal to its latent heat (for phase change) plus the energy to cool it to the phase change temp. So if Model T has a higher freezing point, cooling from room temp to freezing point requires less energy, and then the latent heat: but if the total energy removed is the same, maybe Model T's cooling plus latent heat equals the energy removed, while Model S's cooling plus latent heat is more than the energy removed. So Model T changes phase.
Yes, that makes sense. So the correct option is the first one? Wait no, the second option for Model T says "The Model T's substance changed phase because the strong attraction between the molecules made their movement slower. Its molecules now move in place." No, the energy removal makes movement slower, not the attraction. The first option says "The Model T's substance changed phase because the attraction of the molecules was able to overcome their slower movement. Its molecules now move in place." That's correct: when molecules slow down (from energy removal), the strong attraction can overcome their reduced kinetic energy, holding them in fixed positions (…