QUESTION IMAGE
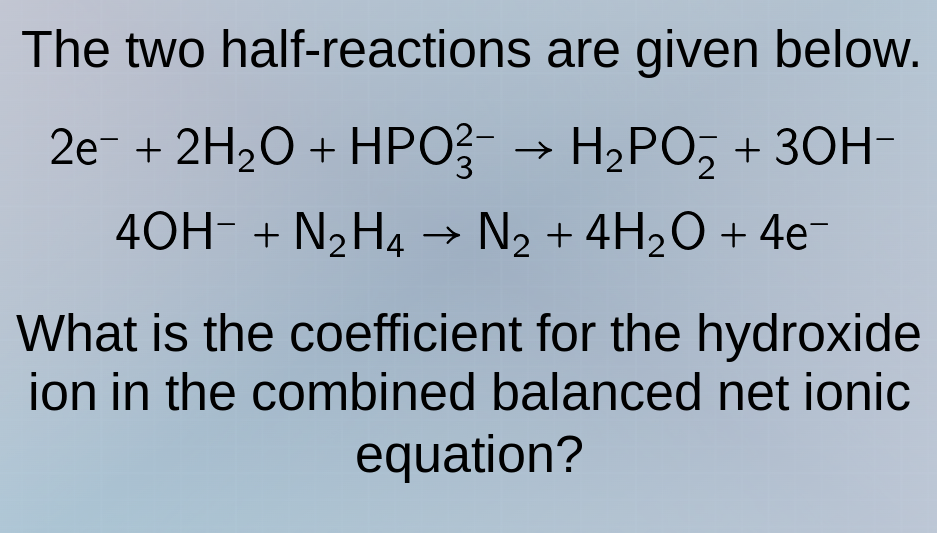
Question
the two half-reactions are given below.
$2e^- + 2h_2o + hpo_3^{2-} \
ightarrow h_2po_2^- + 3oh^-$
$4oh^- + n_2h_4 \
ightarrow n_2 + 4h_2o + 4e^-$
what is the coefficient for the hydroxide ion in the combined balanced net ionic equation?
Step1: Balance electrons in both half - reactions
The first half - reaction: \(2e^-+2H_2O + HPO_3^{2 - }
ightarrow H_2PO_2^-+3OH^-\) has 2 electrons gained.
The second half - reaction: \(4OH^-+N_2H_4
ightarrow N_2 + 4H_2O+4e^-\) has 4 electrons lost.
To balance the number of electrons, we multiply the first half - reaction by 2.
After multiplying the first half - reaction by 2: \(4e^- + 4H_2O+2HPO_3^{2 - }
ightarrow 2H_2PO_2^-+6OH^-\)
Step2: Add the two half - reactions together
Now we add the modified first half - reaction and the second half - reaction:
\((4e^- + 4H_2O+2HPO_3^{2 - })+(4OH^-+N_2H_4)
ightarrow(2H_2PO_2^-+6OH^-)+(N_2 + 4H_2O+4e^-)\)
Step3: Cancel out common species
- Cancel out the \(4e^-\) on both sides.
- Cancel out \(4H_2O\) from both sides (4\(H_2O\) on the left and 4\(H_2O\) on the right).
After cancellation, we get: \(2HPO_3^{2 - }+N_2H_4 + 4OH^-
ightarrow 2H_2PO_2^-+N_2+6OH^-\)
Step4: Simplify the hydroxide ions
Subtract 4\(OH^-\) from both sides: \(2HPO_3^{2 - }+N_2H_4
ightarrow 2H_2PO_2^-+N_2 + 2OH^-\)
Wait, let's re - check the addition:
First half - reaction (after multiplying by 2): \(4e^-+4H_2O + 2HPO_3^{2-}
ightarrow2H_2PO_2^- + 6OH^-\)
Second half - reaction: \(4OH^-+N_2H_4
ightarrow N_2+4H_2O + 4e^-\)
Adding them:
Left side: \(4e^-+4H_2O + 2HPO_3^{2-}+4OH^-+N_2H_4\)
Right side: \(2H_2PO_2^- + 6OH^-+N_2+4H_2O + 4e^-\)
Cancel \(4e^-\) and \(4H_2O\):
Left: \(2HPO_3^{2-}+4OH^-+N_2H_4\)
Right: \(2H_2PO_2^- + 6OH^-+N_2\)
Now, subtract \(4OH^-\) from both sides:
Left: \(2HPO_3^{2-}+N_2H_4\)
Right: \(2H_2PO_2^- + 2OH^-+N_2\)
Wait, maybe I made a mistake in the first multiplication. Let's do it again.
First half - reaction: gain of 2 \(e^-\)
Second half - reaction: loss of 4 \(e^-\)
So multiply the first half - reaction by 2 to make electrons equal (2*2 = 4 electrons gained).
First half - reaction (multiplied by 2): \(4e^-+4H_2O+2HPO_3^{2 - }
ightarrow2H_2PO_2^-+6OH^-\)
Second half - reaction: \(4OH^-+N_2H_4
ightarrow N_2+4H_2O + 4e^-\)
Now add:
\(4e^-+4H_2O + 2HPO_3^{2-}+4OH^-+N_2H_4=2H_2PO_2^-+6OH^-+N_2+4H_2O + 4e^-\)
Cancel \(4e^-\) and \(4H_2O\):
\(2HPO_3^{2-}+4OH^-+N_2H_4=2H_2PO_2^-+6OH^-+N_2\)
Now, subtract \(4OH^-\) from both sides:
\(2HPO_3^{2-}+N_2H_4=2H_2PO_2^-+2OH^-+N_2\)
Wait, no, the question is about the coefficient of \(OH^-\) in the combined balanced equation. Let's check the number of \(OH^-\) on the right and left after canceling.
Wait, another way:
First, find the number of electrons:
Reaction 1: reduction (gain 2 \(e^-\)): \(2e^-+2H_2O + HPO_3^{2-}
ightarrow H_2PO_2^-+3OH^-\)
Reaction 2: oxidation (lose 4 \(e^-\)): \(4OH^-+N_2H_4
ightarrow N_2+4H_2O + 4e^-\)
To balance electrons, multiply reaction 1 by 2:
Reaction 1 (×2): \(4e^-+4H_2O + 2HPO_3^{2-}
ightarrow 2H_2PO_2^-+6OH^-\)
Now add reaction 1 (×2) and reaction 2:
\(4e^-+4H_2O + 2HPO_3^{2-}+4OH^-+N_2H_4
ightarrow 2H_2PO_2^-+6OH^-+N_2+4H_2O + 4e^-\)
Now, cancel \(4e^-\), \(4H_2O\):
Left: \(2HPO_3^{2-}+4OH^-+N_2H_4\)
Right: \(2H_2PO_2^-+6OH^-+N_2\)
Now, the number of \(OH^-\) on the right is 6, on the left is 4. The net \(OH^-\) on the right is \(6 - 4=2\)? Wait, no, when we combine, we have to look at the final equation. Wait, maybe I messed up the direction. Let's do the addition again.
The left side has \(4OH^-\) and the right side has \(6OH^-\). So when we combine, the number of \(OH^-\) in the balanced equation is \(6 - 4 = 2\)? No, wait, let's write the overall equation:
After canceling electrons and water:
\(2HPO_3^{2-}+N_2H_4+4OH^-
ightarrow2H_2PO_2^-+N_2+6OH^-\)
Now, subtract \…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2