QUESTION IMAGE
Question
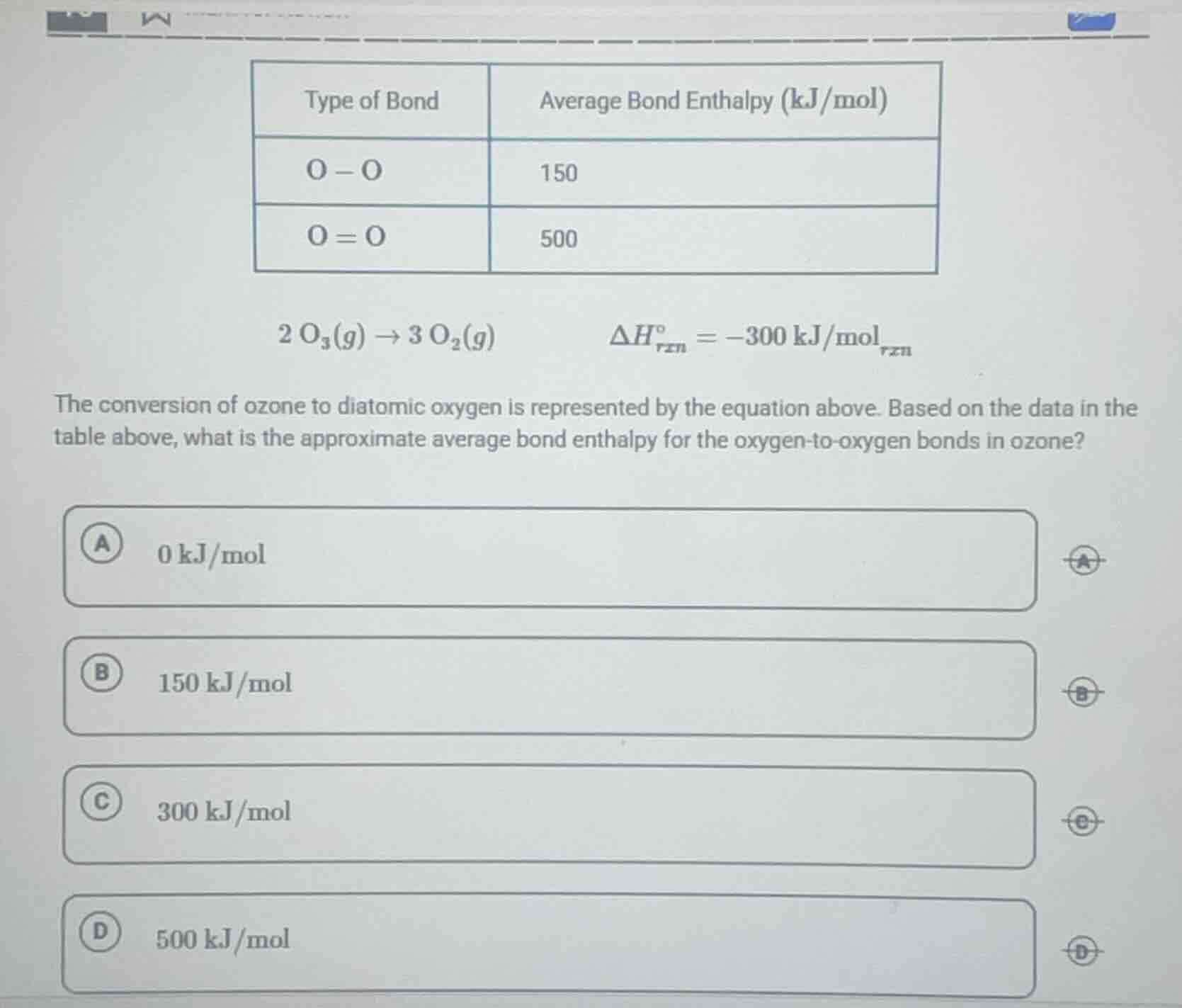
type of bond | average bond enthalpy (kj/mol)
o - o | 150
o = o | 500
2 o₃(g) → 3 o₂(g) δh°_rxn = -300 kj/mol_rxn
the conversion of ozone to diatomic oxygen is represented by the equation above. based on the data in the table above, what is the approximate average bond enthalpy for the oxygen - to - oxygen bonds in ozone?
a 0 kj/mol
b 150 kj/mol
c 300 kj/mol
d 500 kj/mol
Step1: Recall bond enthalpy formula
The enthalpy change of a reaction ($\Delta H_{rxn}$) is calculated as the sum of the bond enthalpies of the reactants minus the sum of the bond enthalpies of the products. Mathematically, $\Delta H_{rxn} = \sum \text{Bond Enthalpies (Reactants)} - \sum \text{Bond Enthalpies (Products)}$.
Step2: Analyze the reaction and bonds
For the reaction $2 \text{O}_3(g)
ightarrow 3 \text{O}_2(g)$:
- Reactants: 2 moles of $\text{O}_3$. Each $\text{O}_3$ molecule has 2 O - O bonds (since the structure of $\text{O}_3$ is O - O - O, so 2 bonds per molecule). So total bonds in reactants: $2 \times 2 = 4$ O - O bonds (let the bond enthalpy of O - O in $\text{O}_3$ be $x$ kJ/mol). So total bond enthalpy of reactants: $4x$.
- Products: 3 moles of $\text{O}_2$. Each $\text{O}_2$ molecule has 1 O = O bond with bond enthalpy 500 kJ/mol. So total bond enthalpy of products: $3 \times 500 = 1500$ kJ/mol.
Step3: Substitute into the formula
We know $\Delta H_{rxn} = - 300$ kJ/mol. Using the formula:
$$-300 = 4x - 1500$$
Step4: Solve for $x$
Add 1500 to both sides:
$$4x = -300 + 1500 = 1200$$
Divide both sides by 4:
$$x = \frac{1200}{4} = 300$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. 300 kJ/mol