QUESTION IMAGE
Question
types of reactions
quiz complete
1 2 3 4 5 6
time remaining
47:32
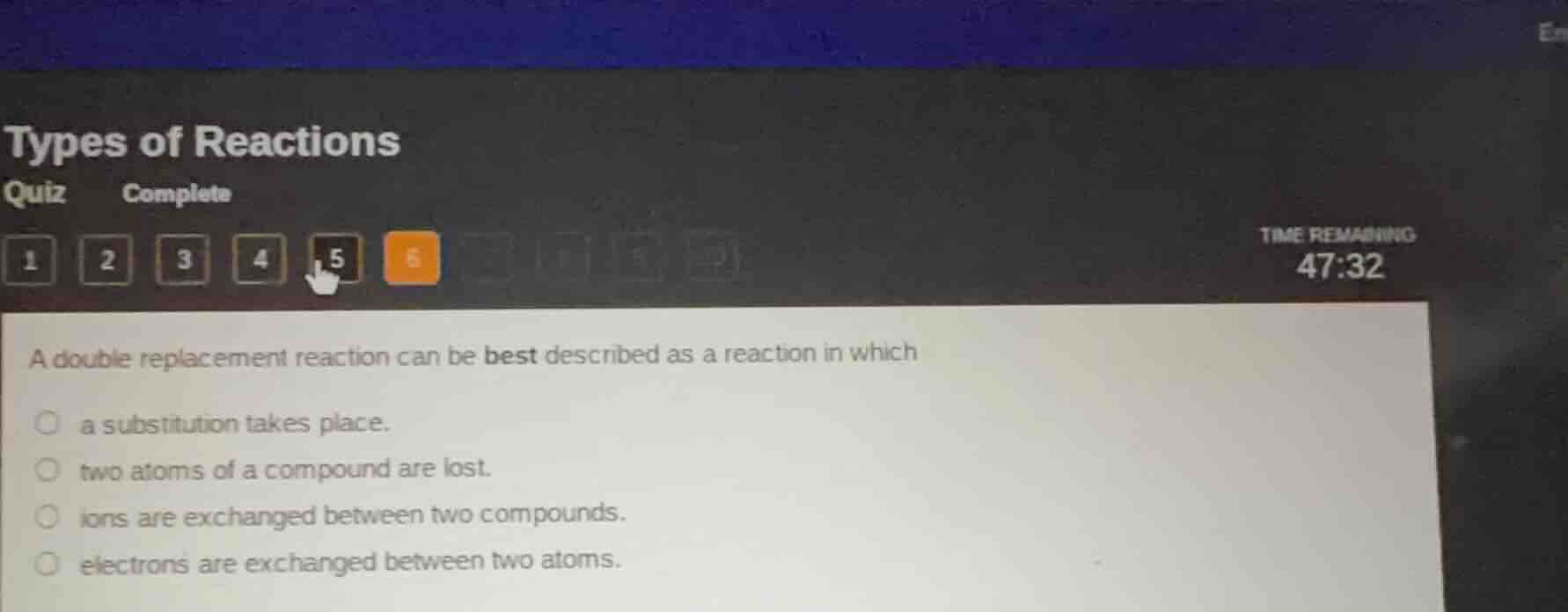
a double replacement reaction can be best described as a reaction in which
a substitution takes place.
two atoms of a compound are lost.
ions are exchanged between two compounds.
electrons are exchanged between two atoms.
A double replacement reaction is defined as a chemical reaction where the positive and negative ions of two ionic compounds swap places, forming new compounds. Substitution is characteristic of single replacement reactions, losing atoms is a decomposition reaction trait, and electron exchange describes redox reactions.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
ions are exchanged between two compounds.