QUESTION IMAGE
Question
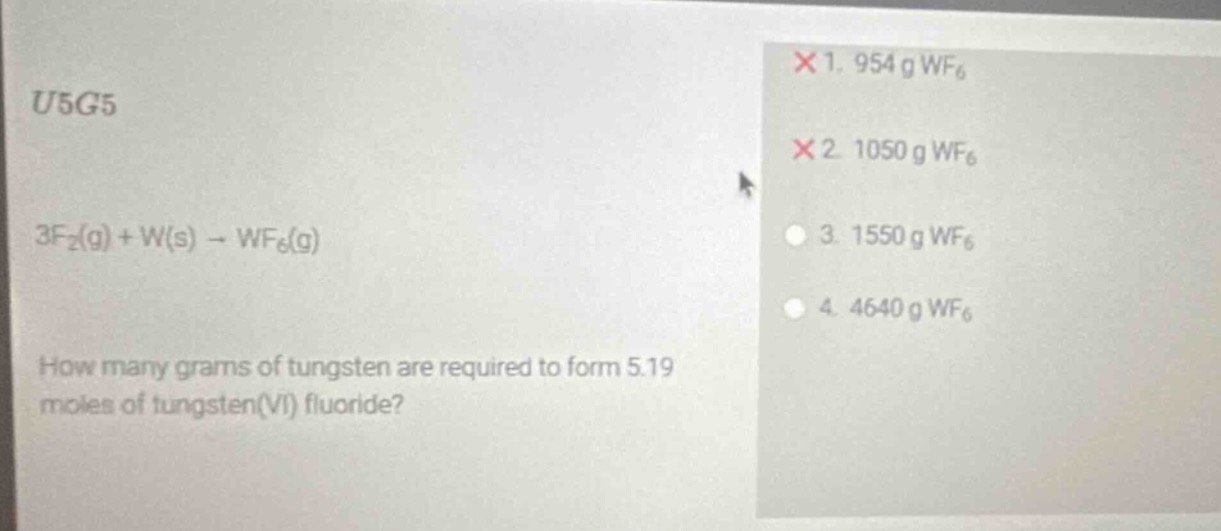
u5g5
3f₂(g) + w(s) → wf₆(g)
how many grams of tungsten are required to form 5.19 moles of tungsten(vi) fluoride?
× 1. 954 g wf₆
× 2. 1050 g wf₆
○ 3. 1550 g wf₆
○ 4. 4640 g wf₆
Step1: Determine the mole ratio
From the balanced chemical equation \(3\text{F}_2(\text{g}) + \text{W}(\text{s})
ightarrow \text{WF}_6(\text{g})\), the mole ratio of \(\text{W}\) to \(\text{WF}_6\) is \(1:1\). So, moles of \(\text{W}\) required = moles of \(\text{WF}_6\) formed = \(5.19\) moles.
Step2: Calculate molar mass of W
The molar mass of tungsten (W) is approximately \(183.84\) g/mol.
Step3: Calculate mass of W
Using the formula \( \text{mass} = \text{moles} \times \text{molar mass} \), we substitute the values:
\(\text{mass of W} = 5.19 \, \text{mol} \times 183.84 \, \text{g/mol}\)
\(\text{mass of W} \approx 5.19 \times 183.84 \approx 954\) g? Wait, no, wait. Wait, the question is about grams of tungsten to form 5.19 moles of \(WF_6\). Wait, maybe I misread. Wait, no, the options are in grams of \(WF_6\)? Wait, no, the question is "How many grams of tungsten are required to form 5.19 moles of tungsten(VI) fluoride?" Wait, but the options are grams of \(WF_6\). Wait, maybe there's a mistake. Wait, no, let's re-express.
Wait, the balanced equation: 1 mole of W produces 1 mole of \(WF_6\). So moles of W = moles of \(WF_6\) = 5.19 mol.
Molar mass of W is 183.84 g/mol.
So mass of W = 5.19 mol 183.84 g/mol ≈ 5.19 183.84 ≈ let's calculate:
5 * 183.84 = 919.2
0.19 * 183.84 ≈ 34.93
Total ≈ 919.2 + 34.93 ≈ 954.13 g. Wait, but the first option is 954 g \(WF_6\)? No, wait, the question is about grams of tungsten, but the options are grams of \(WF_6\). Wait, maybe the question was miswritten, or the options are for a different question. Wait, no, maybe I made a mistake. Wait, the user's question: "How many grams of tungsten are required to form 5.19 moles of tungsten(VI) fluoride?" But the options are grams of \(WF_6\). Wait, perhaps the original question was about grams of \(WF_6\) from tungsten, but the user's text is mixed. Wait, no, let's check the options. The options are 954 g \(WF_6\), 1050 g \(WF_6\), 1550 g \(WF_6\), 4640 g \(WF_6\). Wait, maybe the question is "How many grams of \(WF_6\) are formed from...", but the user wrote "How many grams of tungsten are required...". Wait, maybe it's a typo. Alternatively, maybe the molar mass of \(WF_6\) is calculated. Let's calculate molar mass of \(WF_6\): W is 183.84, F is 19.00, so 183.84 + 619.00 = 183.84 + 114 = 297.84 g/mol. Then mass of \(WF_6\) = 5.19 mol 297.84 g/mol ≈ 5.19 * 297.84 ≈ let's calculate:
5 * 297.84 = 1489.2
0.19 * 297.84 ≈ 56.59
Total ≈ 1489.2 + 56.59 ≈ 1545.79 ≈ 1550 g. Ah, so maybe the question was supposed to be about grams of \(WF_6\) formed from 5.19 moles of W, or the user mixed up. But given the options, 1550 g \(WF_6\) is the answer. So let's redo:
If the question is "How many grams of \(WF_6\) are formed from 5.19 moles of W?" (since mole ratio W:WF6 is 1:1), then:
Molar mass of \(WF_6\) = 183.84 + 6*19.00 = 183.84 + 114 = 297.84 g/mol.
Mass of \(WF_6\) = 5.19 mol 297.84 g/mol ≈ 5.19 297.84 ≈ 1550 g (rounded). So the correct option is 3. 1550 g \(WF_6\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- 1550 g \(WF_6\)