QUESTION IMAGE
Question
unit review - matter and change
skip
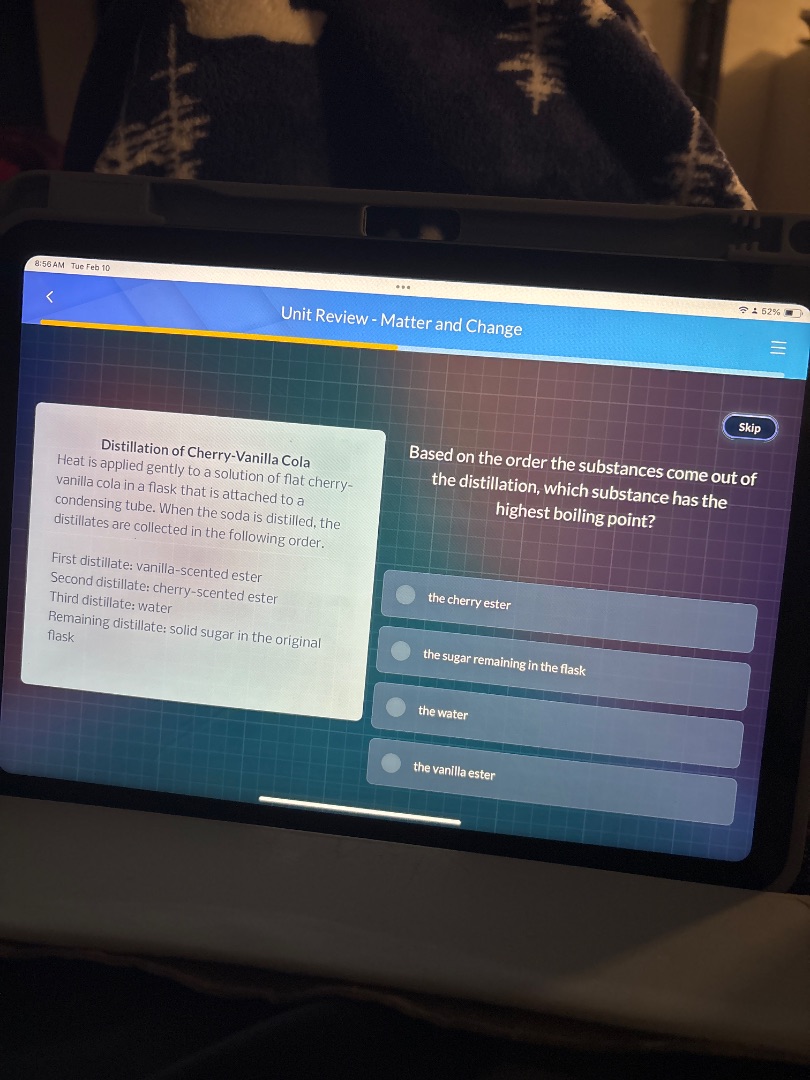
distillation of cherry-vanilla cola
heat is applied gently to a solution of flat cherry-vanilla cola in a flask that is attached to a condensing tube. when the soda is distilled, the distillates are collected in the following order.
first distillate: vanilla-scented ester
second distillate: cherry-scented ester
third distillate: water
remaining distillate: solid sugar in the original flask
based on the order the substances come out of the distillation, which substance has the highest boiling point?
the cherry ester
the sugar remaining in the flask
the water
the vanilla ester
In distillation, substances with lower boiling points vaporize and are collected first, while those with higher boiling points remain longer and are collected last (or stay in the flask). The solid sugar is the last remaining component, meaning it has the highest boiling point among the listed substances.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
the sugar remaining in the flask