QUESTION IMAGE
Question
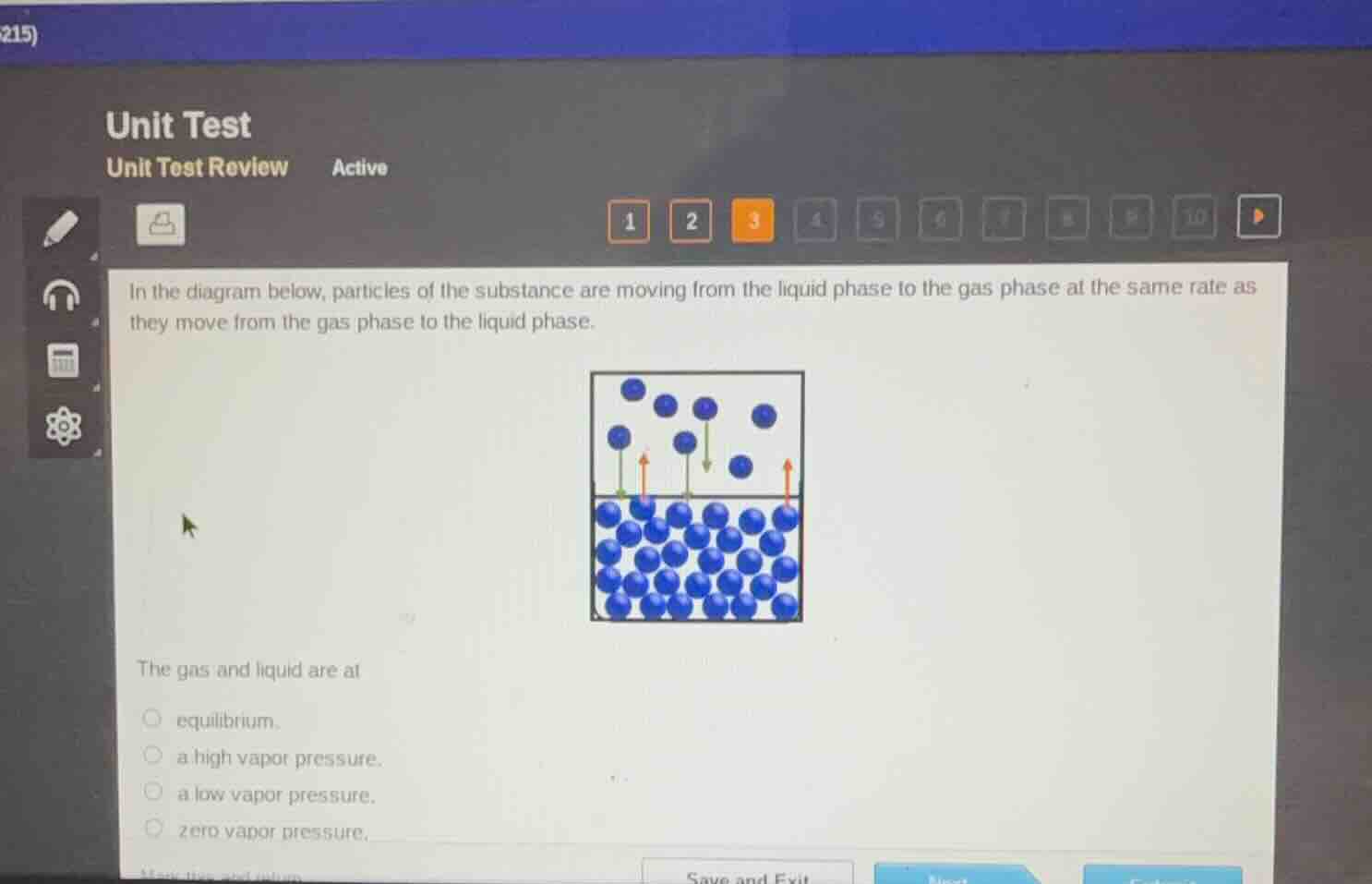
unit test unit test review active 1 2 3 4 5 6 7 8 9 10 in the diagram below, particles of the substance are moving from the liquid phase to the gas phase at the same rate as they move from the gas phase to the liquid phase. the gas and liquid are at equilibrium. a high vapor pressure. a low vapor pressure. zero vapor pressure. save and exit
When the rate of particles moving from liquid to gas equals the rate of particles moving from gas to liquid, the system has reached a state where the properties of the liquid and gas phases remain stable. This is the definition of phase equilibrium for a liquid-vapor system. Vapor pressure refers to the pressure of the gas phase at this state, but the key description here matches the equilibrium condition, not just a high/low/zero vapor pressure value.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
equilibrium.