QUESTION IMAGE
Question
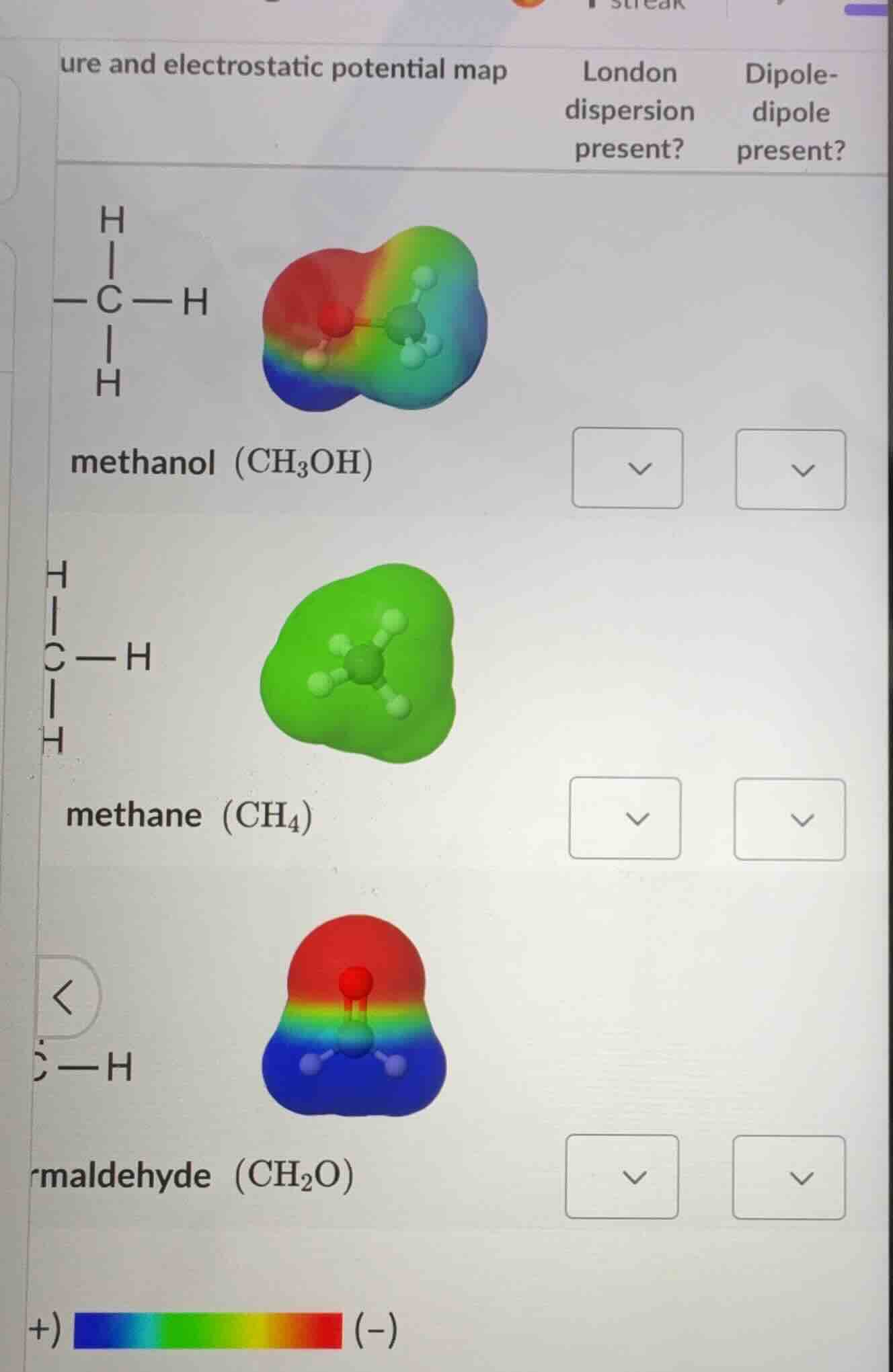
ure and electrostatic potential map london dispersion present? dipole-dipole present? methanol (ch₃oh) methane (ch₄) maldehyde (ch₂o) +) (-)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
methanol (CH₃OH): London dispersion present? Yes; Dipole-dipole present? Yes
methane (CH₄): London dispersion present? Yes; Dipole-dipole present? No
formaldehyde (CH₂O): London dispersion present? Yes; Dipole-dipole present? Yes