QUESTION IMAGE
Question
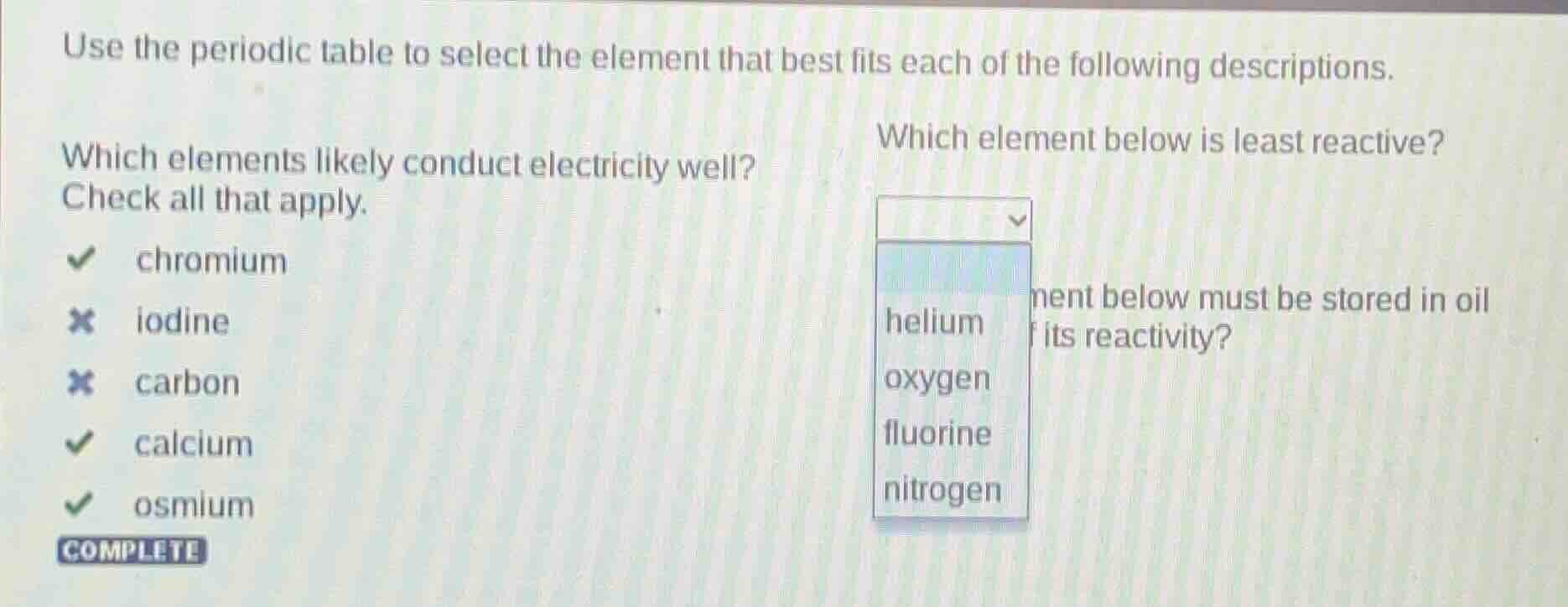
use the periodic table to select the element that best fits each of the following descriptions. which elements likely conduct electricity well? check all that apply. chromium iodine carbon calcium osmium complete which element below is least reactive? helium oxygen fluorine nitrogen ment below must be stored in oil its reactivity?
Sub - question 1: Which elements likely conduct electricity well?
Metals are good conductors of electricity. Chromium, calcium, and osmium are metals. Iodine is a non - metal (halogen) and carbon (in its common forms like graphite is a conductor but in diamond is not, and the general classification here likely considers carbon as a non - metal conductor in the context of this question's options) is not a typical metal conductor. So chromium, calcium, and osmium conduct electricity well.
Among the given elements (helium, oxygen, fluorine, nitrogen), helium is a noble gas. Noble gases have a full valence electron shell, which makes them extremely stable and least reactive compared to other elements. Oxygen is a reactive non - metal, fluorine is one of the most reactive non - metals, and nitrogen is relatively reactive but not as stable as helium.
Elements that are highly reactive and react with air or moisture are stored in oil. Alkali metals (like sodium, potassium) or alkaline earth metals (like calcium in some cases but more reactive ones) are stored in oil. But among common elements, sodium, potassium are typical, but if we consider the context, if we assume the options (not fully visible) but generally, elements like sodium (Na) or potassium (K) are stored in oil. If we consider the previous options, maybe a highly reactive metal. But from the visible part, if we assume the question is about a reactive metal, for example, if we consider the elements, a metal like sodium (if in options) or potassium. But based on the given elements in the second question, if we assume the third question's options might include a reactive metal. However, from the current image, if we have to answer based on general knowledge, highly reactive metals (Group 1 or 2) are stored in oil. For example, sodium is stored in oil because it reacts vigorously with air and water.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
chromium, calcium, osmium