QUESTION IMAGE
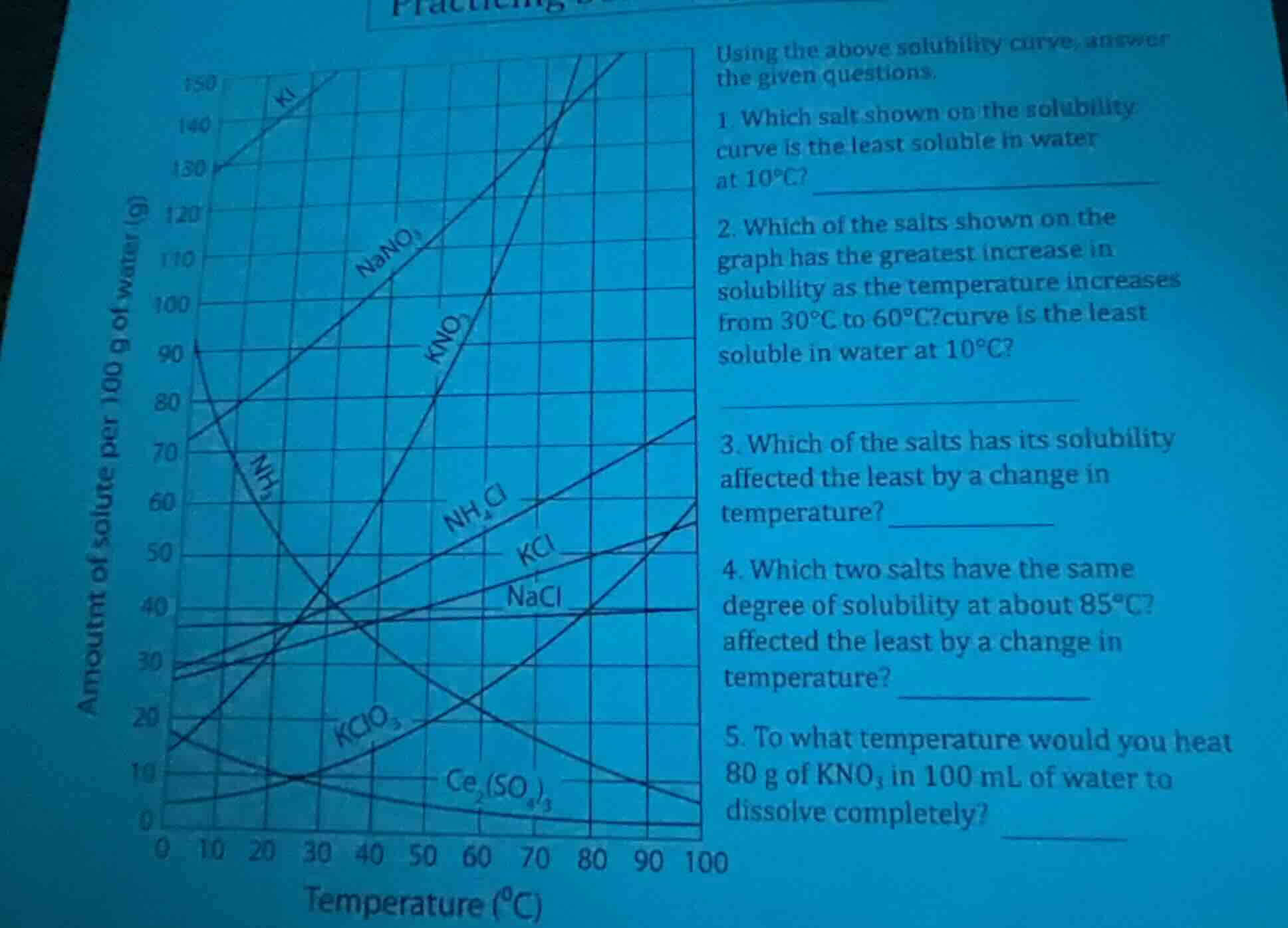
Question
using the above solubility curve, answer the given questions. 1. which salt shown on the solubility curve is the least soluble in water at 10°c? 2. which of the salts shown on the graph has the greatest increase in solubility as the temperature increases from 30°c to 60°c? curve is the least soluble in water at 10°c? 3. which of the salts has its solubility affected the least by a change in temperature? 4. which two salts have the same degree of solubility at about 85°c? affected the least by a change in temperature? 5. to what temperature would you heat 80 g of kno₃ in 100 ml of water to dissolve completely?
Question 1
To determine the least soluble salt at \(10^\circ\text{C}\), we examine the solubility curve. At \(10^\circ\text{C}\), the salt with the lowest solubility (lowest y - value on the curve) is \(KClO_3\) (or potentially other low - solubility salts at this temperature, but from the curve, \(KClO_3\) has a very low solubility at \(10^\circ\text{C}\) compared to others like \(KNO_3\), \(KI\), etc.).
To find the salt with the greatest increase in solubility from \(30^\circ\text{C}\) to \(60^\circ\text{C}\), we calculate the difference in solubility (y - axis values) for each salt between these two temperatures. For \(KNO_3\), the solubility at \(30^\circ\text{C}\) is relatively low and at \(60^\circ\text{C}\) is much higher. The slope of the solubility curve for \(KNO_3\) between \(30^\circ\text{C}\) and \(60^\circ\text{C}\) is steeper than for other salts, indicating a larger increase in solubility.
A salt whose solubility is affected the least by temperature change has a nearly flat solubility curve. \(NaCl\) has a solubility curve that changes very little as temperature varies. By comparing the slopes of the solubility curves of all salts, \(NaCl\)'s curve has the smallest slope (least change in solubility with temperature).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(KClO_3\) (or other correct salt based on the curve, but \(KClO_3\) is a common answer here)