QUESTION IMAGE
Question
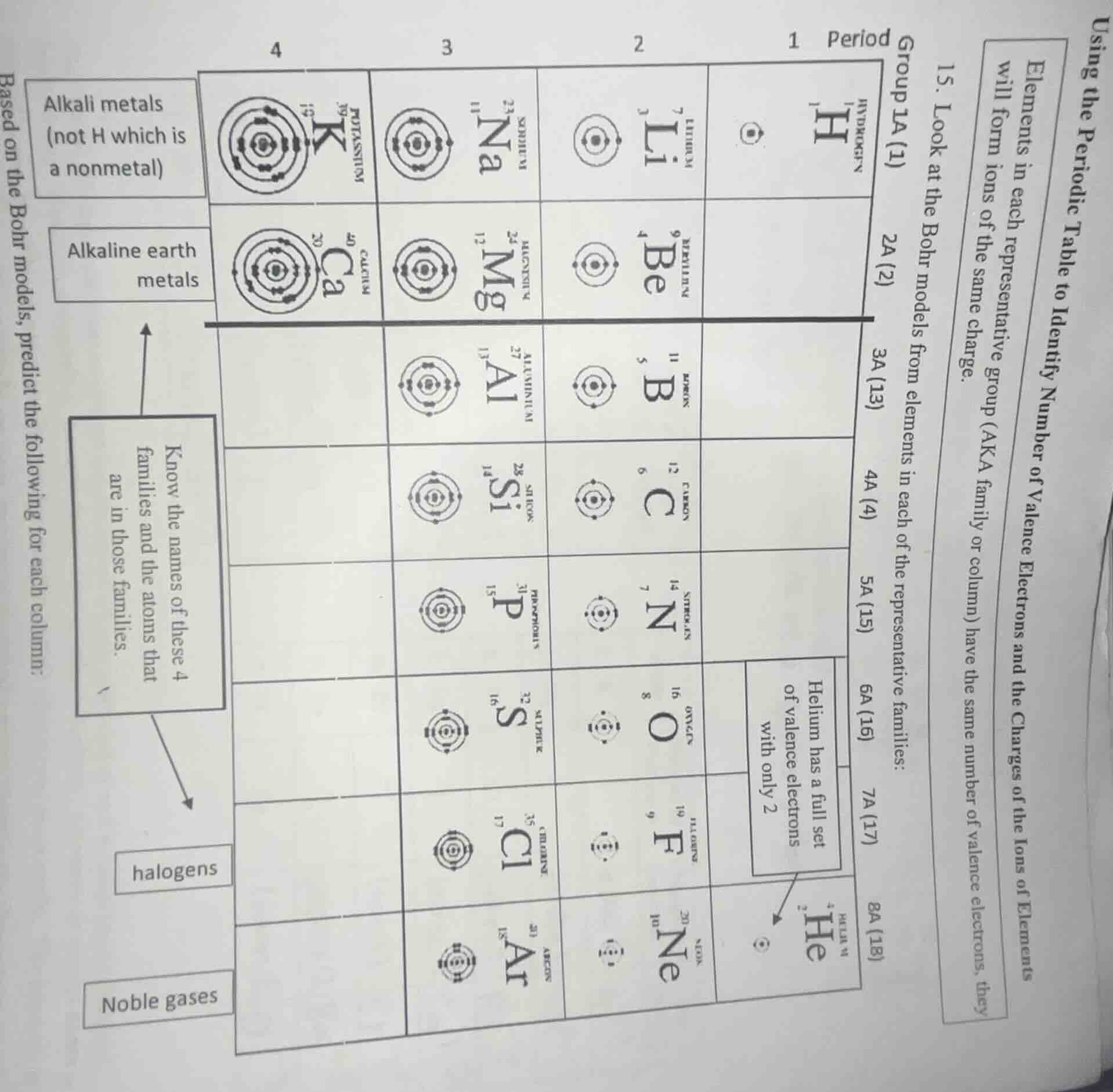
using the periodic table to identify number of valence electrons and the charges of the ions of elements
elements in each representative group (aka family or column) have the same number of valence electrons, they will form ions of the same charge.
- look at the bohr models from elements in each of the representative families:
based on the bohr models, predict the following for each column:
(the chart contains elements symbols, atomic numbers, mass numbers, and bohr models for elements in different groups and periods, along with labels for alkali metals, alkaline earth metals, halogens, noble gases, and descriptions like \alkali metals (not h which is a nonmetal)\, \helium has a full set of valence electrons with only 2\)
To solve this, we analyze each group:
Group 1A (1) (Alkali Metals: H is nonmetal, others are metals)
- Valence Electrons: 1 (since group number 1A means 1 valence electron).
- Ion Charge: These metals tend to lose 1 electron to achieve a stable octet (or duet for Li, Na, K, etc.). So they form cations with a charge of \( +1 \) (e.g., \( \text{Li}^+ \), \( \text{Na}^+ \), \( \text{K}^+ \); H is nonmetal, so it behaves differently, but the metal members of 1A form \( +1 \) ions).
Group 2A (2) (Alkaline Earth Metals)
- Valence Electrons: 2 (group number 2A means 2 valence electrons).
- Ion Charge: These metals lose 2 electrons to achieve stability, forming cations with a charge of \( +2 \) (e.g., \( \text{Be}^{2+} \), \( \text{Mg}^{2+} \), \( \text{Ca}^{2+} \)).
Group 3A (13)
- Valence Electrons: 3 (group number 13, so 3 valence electrons: \( 13 - 10 = 3 \) for main - group elements).
- Ion Charge: These elements (mostly metals like Al, B is metalloid) tend to lose 3 electrons, forming cations with a charge of \( +3 \) (e.g., \( \text{Al}^{3+} \), \( \text{B}^{3+} \) in ionic compounds).
Group 4A (14)
- Valence Electrons: 4 (group number 14, \( 14 - 10 = 4 \) valence electrons).
- Ion Charge: Elements here (like C, Si) can either lose 4 electrons (forming \( +4 \) cations) or gain 4 electrons (forming \( -4 \) anions), but they often form covalent bonds. However, for ionic behavior, metals (less common here) would form \( +4 \) and non - metals \( -4 \), but the typical charge trend is \( \pm 4 \) (more likely to share electrons in many cases).
Group 5A (15)
- Valence Electrons: 5 (group number 15, \( 15 - 10 = 5 \) valence electrons).
- Ion Charge: These non - metals (like N, P) tend to gain 3 electrons to achieve a stable octet, forming anions with a charge of \( -3 \) (e.g., \( \text{N}^{3 - } \), \( \text{P}^{3 - } \)).
Group 6A (16)
- Valence Electrons: 6 (group number 16, \( 16 - 10 = 6 \) valence electrons).
- Ion Charge: Non - metals here (like O, S) gain 2 electrons to achieve stability, forming anions with a charge of \( -2 \) (e.g., \( \text{O}^{2 - } \), \( \text{S}^{2 - } \)).
Group 7A (17) (Halogens)
- Valence Electrons: 7 (group number 17, \( 17 - 10 = 7 \) valence electrons).
- Ion Charge: These non - metals gain 1 electron to achieve a stable octet, forming anions with a charge of \( -1 \) (e.g., \( \text{F}^- \), \( \text{Cl}^- \), \( \text{Br}^- \)).
Group 8A (18) (Noble Gases)
- Valence Electrons: 8 (except He, which has 2). He has 2 valence electrons (a full duet), and other noble gases (Ne, Ar, etc.) have 8 valence electrons (a full octet).
- Ion Charge: These elements have a stable electron configuration, so they rarely form ions (charge is \( 0 \)).
If we take a specific example, say Group 1A (1):
Step 1: Determine Valence Electrons
For Group 1A (main - group elements), the number of valence electrons is equal to the group number. So Group 1A elements have 1 valence electron.
Step 2: Determine Ion Charge
Alkali metals (Group 1A, except H) have 1 valence electron. To achieve a stable electron configuration (like the nearest noble gas), they lose this 1 electron. When an atom loses an electron, it becomes a cation with a charge equal to the number of electrons lost. So they form ions with a charge of \( +1 \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
(for Group 1A as an example):
- Valence Electrons: 1
- Ion Charge: \( +1 \)
(You can follow the same logic for other groups. If you want a specific group's answer, just specify the group, and we can refine it further.)