QUESTION IMAGE
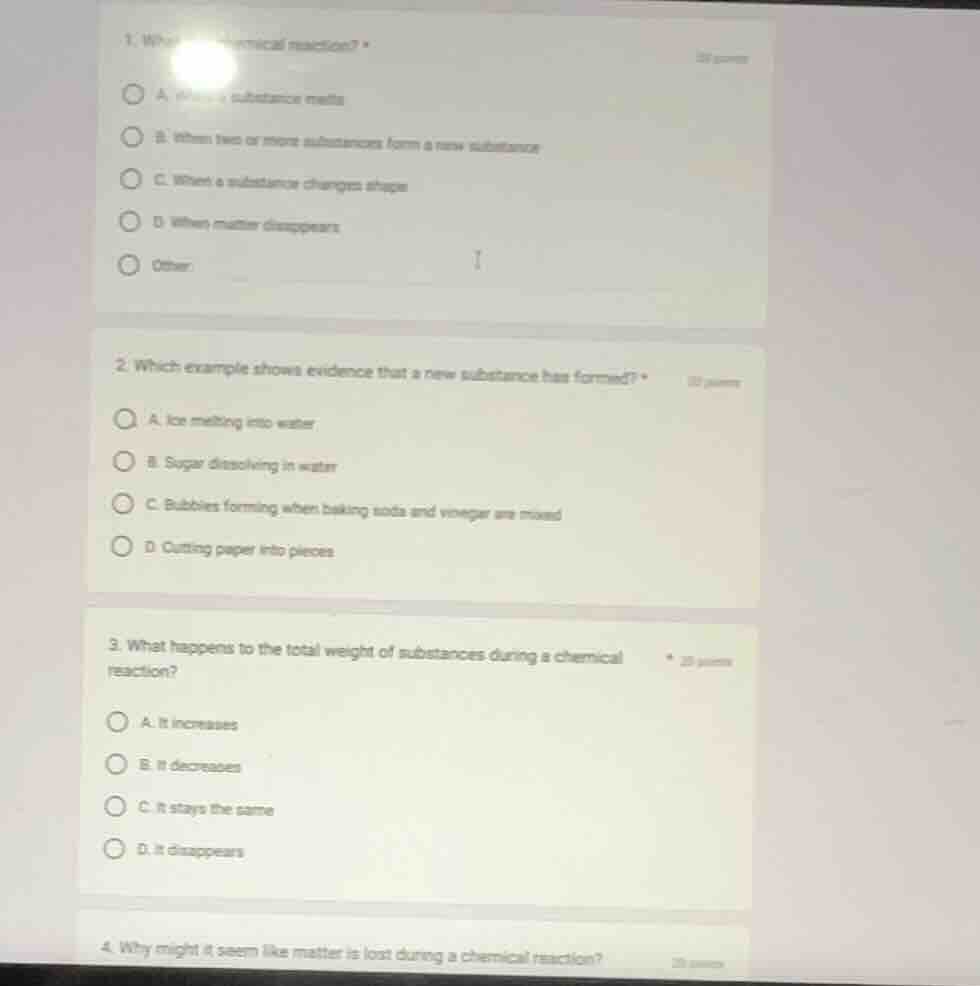
Question
- wh... chemical reaction? *
a. when a substance melts
b. when two or more substances form a new substance
c. when a substance changes shape
d. when matter disappears
other:
- which example shows evidence that a new substance has formed? *
a. ice melting into water
b. sugar dissolving in water
c. bubbles forming when baking soda and vinegar are mixed
d. cutting paper into pieces
- what happens to the total weight of substances during a chemical reaction? *
a. it increases
b. it decreases
c. it stays the same
d. it disappears
- why might it seem like matter is lost during a chemical reaction?
Brief Explanations
- A chemical reaction involves transforming reactants into new substances, not just physical changes or matter vanishing.
- Bubbles from baking soda and vinegar indicate a new gas substance, unlike the physical changes in other options.
- The law of conservation of mass states mass is conserved in chemical reactions.
- Matter may seem lost if a gaseous product is released into the air and not measured, but no actual mass is lost.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- B. When two or more substances form a new substance
- C. Bubbles forming when baking soda and vinegar are mixed
- C. It stays the same
- It may seem like matter is lost if a gaseous product from the reaction is released into the atmosphere and not accounted for in measurements, but the total mass of all substances (including the gas) remains unchanged per the law of conservation of mass.