QUESTION IMAGE
Question
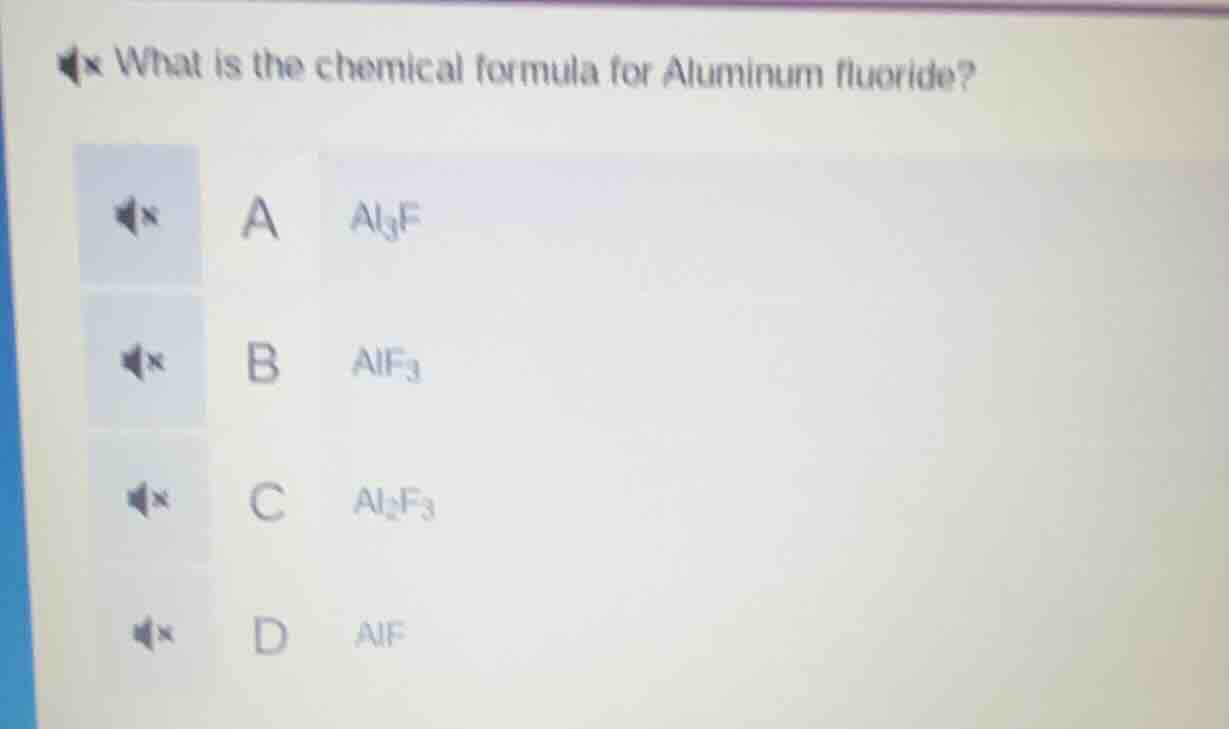
what is the chemical formula for aluminum fluoride? a $\ce{al_{3}f}$ b $\ce{alf_{3}}$ c $\ce{al_{2}f_{3}}$ d $\ce{alf}$
Step1: Identify ion charges
Aluminum forms $\text{Al}^{3+}$; fluorine forms $\text{F}^-$
Step2: Balance charge neutrality
3 $\text{F}^-$ ions balance 1 $\text{Al}^{3+}$ ion
Step3: Write the formula
Combine symbols with subscripts for charge balance
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. $\text{AlF}_3$