QUESTION IMAGE
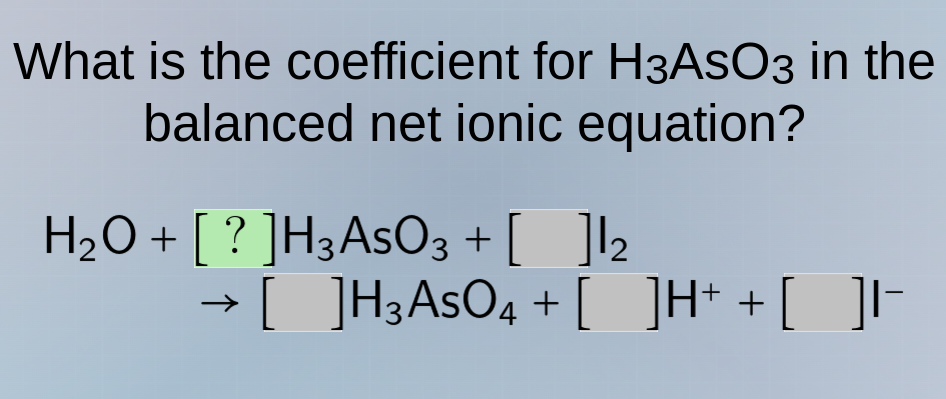
Question
what is the coefficient for h₃aso₃ in the balanced net ionic equation? h₂o + ?h₃aso₃ + i₂ → h₃aso₄ + h⁺ + i⁻
Step1: Identify oxidation states
- For As in \( \ce{H3AsO3} \): Let oxidation state of As be \( x \). \( 3(+1) + x + 3(-2) = 0 \) → \( x = +3 \).
- For As in \( \ce{H3AsO4} \): \( 3(+1) + x + 4(-2) = 0 \) → \( x = +5 \). So As is oxidized (loses 2 electrons).
- For I in \( \ce{I2} \): oxidation state 0; in \( \ce{I-} \): -1. So I is reduced (gains 1 electron per I atom, 2 electrons per \( \ce{I2} \) molecule).
Step2: Balance electrons
- Oxidation: \( \ce{H3AsO3 -> H3AsO4} \), loss of 2 e⁻ per \( \ce{H3AsO3} \).
- Reduction: \( \ce{I2 -> 2I-} \), gain of 2 e⁻ per \( \ce{I2} \). So mole ratio of \( \ce{H3AsO3} \) to \( \ce{I2} \) is 1:1 (to balance electrons: 2 e⁻ lost = 2 e⁻ gained).
Step3: Balance As and I
- Put coefficient 1 for \( \ce{H3AsO3} \) and 1 for \( \ce{I2} \). Now:
- As: 1 on left (in \( \ce{H3AsO3} \)) and 1 on right (in \( \ce{H3AsO4} \)) → balance.
- I: 2 on left (in \( \ce{I2} \)) and 2 on right (in \( \ce{I-} \)) → put 2 for \( \ce{I-} \).
Step4: Balance O and H
- O: Left (in \( \ce{H3AsO3} \)): 3; Right (in \( \ce{H3AsO4} \)): 4. So add 1 \( \ce{H2O} \) on left (provides 1 O). Now O: 3 + 1 = 4 (balanced).
- H: Left: \( \ce{H2O} \) (2 H) + \( \ce{H3AsO3} \) (3 H) = 5 H. Right: \( \ce{H3AsO4} \) (3 H) + \( \ce{H+} \) (let coefficient be \( x \)) + \( \ce{I-} \) (0 H) = 3 + \( x \) H. So \( 5 = 3 + x \) → \( x = 2 \). So \( \ce{H+} \) coefficient is 2.
Now the balanced equation: \( \ce{H2O + H3AsO3 + I2 -> H3AsO4 + 2H+ + 2I-} \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1