QUESTION IMAGE
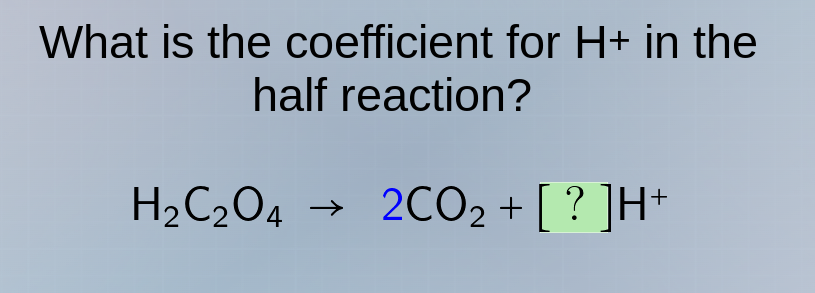
Question
what is the coefficient for h+ in the half reaction? h₂c₂o₄ → 2co₂ + ?h⁺
Step1: Analyze the number of H atoms on each side
On the left side, in \(H_2C_2O_4\), there are 2 H atoms. On the right side, the H atoms are in \(H^+\).
Step2: Balance the H atoms
To balance the H atoms, since there are 2 H atoms in \(H_2C_2O_4\), we need 2 \(H^+\) ions on the right side to have the same number of H atoms. So the coefficient for \(H^+\) should be 2.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2