QUESTION IMAGE
Question
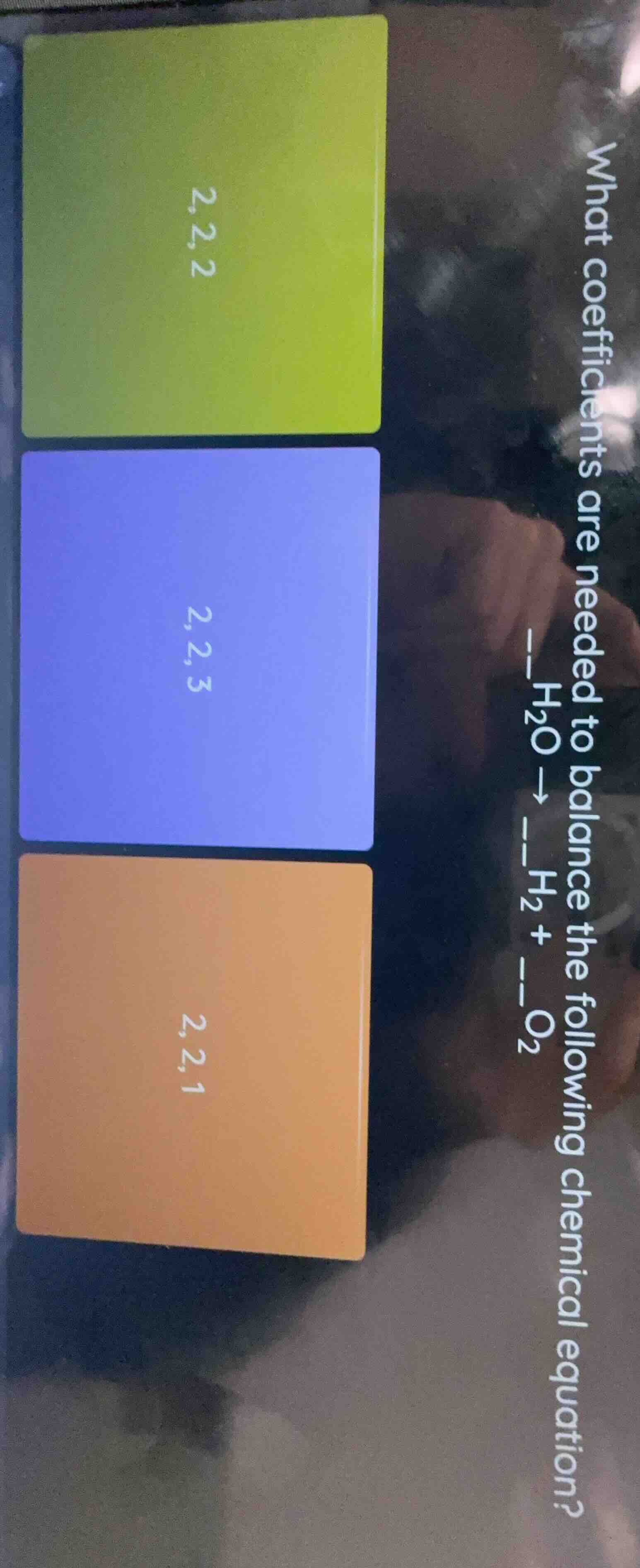
what coefficients are needed to balance the following chemical equation? h₂o → h₂ + __ o₂ options: 2,2,2; 2,2,3; 2,2,1
Step1: Analyze the chemical equation
The unbalanced equation is \( \text{H}_2\text{O}
ightarrow \text{H}_2 + \text{O}_2 \). We need to balance the number of hydrogen and oxygen atoms on both sides.
Step2: Balance hydrogen atoms
On the left side, each \( \text{H}_2\text{O} \) has 2 H atoms. On the right side, \( \text{H}_2 \) has 2 H atoms. Let's assume the coefficient of \( \text{H}_2\text{O} \) is \( a \), \( \text{H}_2 \) is \( b \), and \( \text{O}_2 \) is \( c \). So for H atoms: \( 2a = 2b \), which simplifies to \( a = b \).
Step3: Balance oxygen atoms
On the left side, each \( \text{H}_2\text{O} \) has 1 O atom. On the right side, \( \text{O}_2 \) has 2 O atoms. So for O atoms: \( a = 2c \).
Step4: Test the options
- Option 2,2,2: Equation becomes \( 2\text{H}_2\text{O}
ightarrow 2\text{H}_2 + 2\text{O}_2 \). Left O: 2, Right O: 4. Not balanced.
- Option 2,2,3: Equation becomes \( 2\text{H}_2\text{O}
ightarrow 2\text{H}_2 + 3\text{O}_2 \). Left O: 2, Right O: 6. Not balanced.
- Option 2,2,1: Equation becomes \( 2\text{H}_2\text{O}
ightarrow 2\text{H}_2 + 1\text{O}_2 \). Left H: 4, Right H: 4; Left O: 2, Right O: 2. Balanced. Wait, no, wait. Wait the correct balanced equation for \( \text{H}_2\text{O} \) decomposition is \( 2\text{H}_2\text{O}
ightarrow 2\text{H}_2 + \text{O}_2 \). Wait, maybe I misread the options. Wait the options are 2,2,2; 2,2,3; 2,2,1? Wait no, looking at the image, the options are 2,2,2 (green), 2,2,3 (blue), 2,2,1 (orange)? Wait no, the equation is \( \_\_ \text{H}_2\text{O}
ightarrow \_\_ \text{H}_2 + \_\_ \text{O}_2 \). Let's balance properly.
Let's do it correctly. The decomposition of water is \( 2\text{H}_2\text{O} \xrightarrow{\text{electrolysis}} 2\text{H}_2 + \text{O}_2 \). So coefficients are 2 (for \( \text{H}_2\text{O} \)), 2 (for \( \text{H}_2 \)), and 1 (for \( \text{O}_2 \)). Wait but the options: the orange one is 2,2,1? Wait the green is 2,2,2; blue 2,2,3; orange 2,2,1? Wait maybe the user made a typo, but let's check again.
Wait the correct balanced equation is \( 2\text{H}_2\text{O} = 2\text{H}_2 + \text{O}_2 \). So coefficients are 2, 2, 1. So the orange option (2,2,1) is correct? Wait no, wait the options as per the image: green is 2,2,2; blue 2,2,3; orange 2,2,1. Wait maybe I messed up. Wait let's check each option:
- 2,2,2: \( 2\text{H}_2\text{O}
ightarrow 2\text{H}_2 + 2\text{O}_2 \). H: 4 on both, O: 2 vs 4. Not balanced.
- 2,2,3: \( 2\text{H}_2\text{O}
ightarrow 2\text{H}_2 + 3\text{O}_2 \). H: 4 on both, O: 2 vs 6. Not balanced.
- 2,2,1: \( 2\text{H}_2\text{O}
ightarrow 2\text{H}_2 + 1\text{O}_2 \). H: 4 on both, O: 2 on both. Balanced. Wait, but the standard equation is \( 2\text{H}_2\text{O}
ightarrow 2\text{H}_2 + \text{O}_2 \), so coefficients 2,2,1. So the orange option (2,2,1) is correct? Wait but maybe the options are written as (coefficient of \( \text{H}_2\text{O} \), coefficient of \( \text{H}_2 \), coefficient of \( \text{O}_2 \)). So 2,2,1.
Wait but maybe I made a mistake. Wait let's re-express:
Unbalanced: \( \text{H}_2\text{O}
ightarrow \text{H}_2 + \text{O}_2 \)
Balance O: Multiply \( \text{H}_2\text{O} \) by 2: \( 2\text{H}_2\text{O}
ightarrow \text{H}_2 + \text{O}_2 \)
Now H: Left has 4, right has 2. Multiply \( \text{H}_2 \) by 2: \( 2\text{H}_2\text{O}
ightarrow 2\text{H}_2 + \text{O}_2 \)
Now O: Left has 2, right has 2. Balanced. So coefficients are 2 (H2O), 2 (H2), 1 (O2). So the option with 2,2,1.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2,2,1 (the orange option)