QUESTION IMAGE
Question
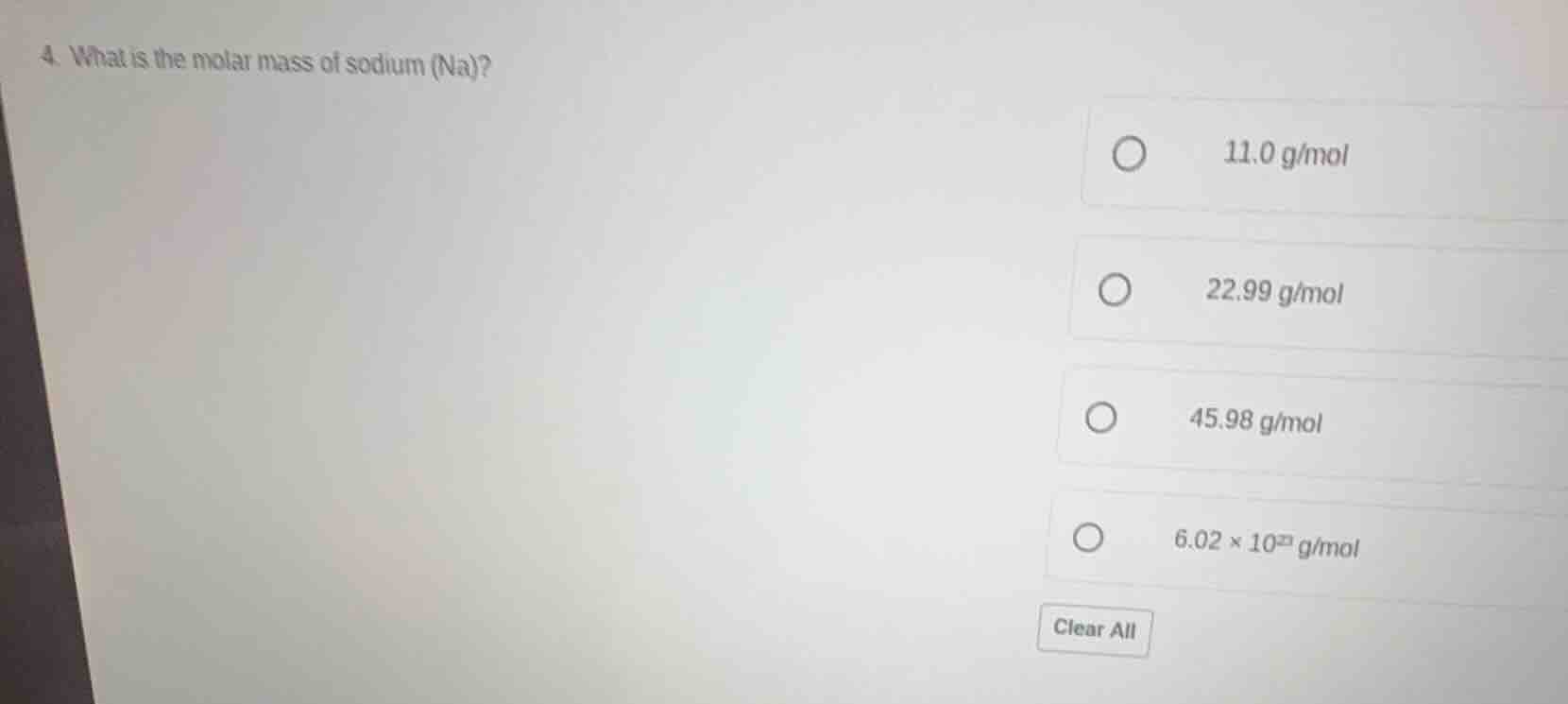
- what is the molar mass of sodium (na)?
11.0 g/mol
22.99 g/mol
45.98 g/mol
6.02 × 10²³ g/mol
clear all
Brief Explanations
The molar mass of an element is numerically equal to its atomic mass (in atomic mass units) and has units of g/mol. The atomic mass of sodium (Na) from the periodic table is approximately 22.99 atomic mass units, so its molar mass is 22.99 g/mol.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. 22.99 g/mol (assuming the second option is labeled B; if the original options had labels like A, B, C, D with the second being B, else adjust the label as per the original but the value is 22.99 g/mol)