QUESTION IMAGE
Question
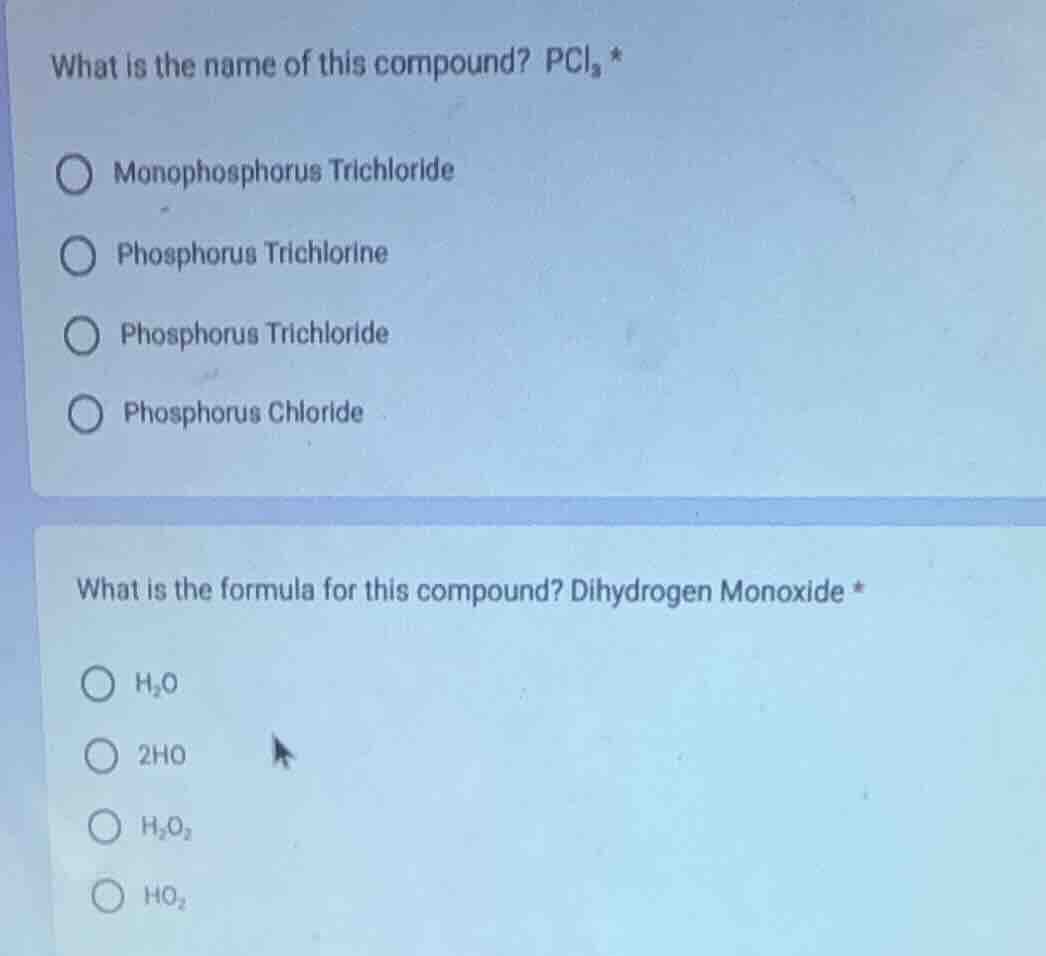
what is the name of this compound? pcl₃ *
monophosphorus trichloride
phosphorus trichlorine
phosphorus trichloride
phosphorus chloride
what is the formula for this compound? dihydrogen monoxide *
h₂o
2ho
h₂o₂
ho₂
First Sub - Question: Naming of \(PCl_3\)
To name a binary molecular compound like \(PCl_3\), we use prefixes to indicate the number of atoms of each element. The first element is named as is, and the second element has its ending changed to -ide, with prefixes (mono - for 1, di - for 2, tri - for 3, etc.). For \(PCl_3\), there is 1 phosphorus (no prefix needed as it's the first element) and 3 chlorines (tri - chlor - ide). So the correct name is Phosphorus Trichloride.
The prefixes in the name of a molecular compound tell us the number of atoms of each element. "Dihydrogen" means 2 hydrogen atoms (\(H_2\)) and "Monoxide" means 1 oxygen atom (O). So combining these, the formula is \(H_2O\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. Phosphorus Trichloride