QUESTION IMAGE
Question
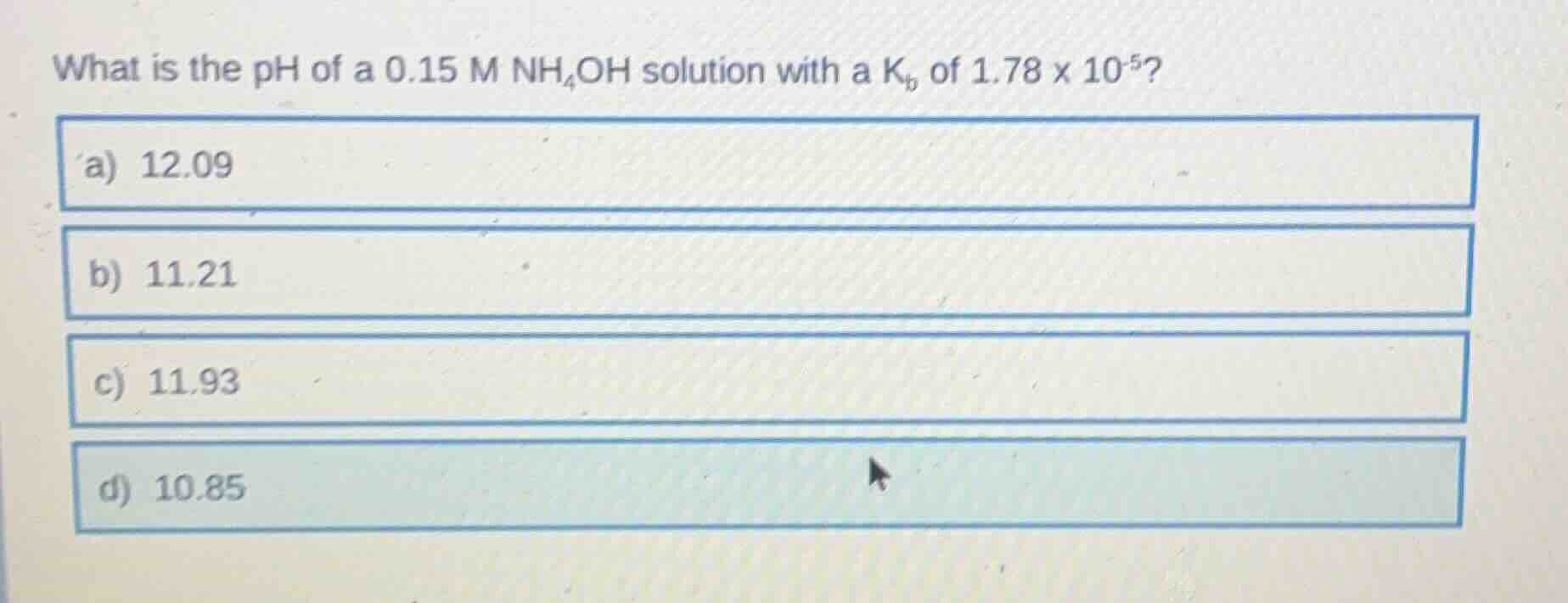
what is the ph of a 0.15 m nh₄oh solution with a kᵦ of 1.78 x 10⁻⁵?
a) 12.09
b) 11.21
c) 11.93
d) 10.85
Step1: Calculate $[OH^-]$
For weak bases, $[OH^-] = \sqrt{K_b \times c}$, where $K_b=1.78 \times 10^{-5}$ and $c=0.15\ \text{M}$.
$$[OH^-] = \sqrt{1.78 \times 10^{-5} \times 0.15}$$
$$[OH^-] = \sqrt{2.67 \times 10^{-6}} \approx 1.634 \times 10^{-3}\ \text{M}$$
Step2: Calculate pOH
$\text{pOH} = -\log[OH^-]$
$$\text{pOH} = -\log(1.634 \times 10^{-3}) \approx 2.787$$
Step3: Calculate pH
Use $\text{pH} + \text{pOH} = 14$
$$\text{pH} = 14 - 2.787 \approx 11.21$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
b) 11.21