QUESTION IMAGE
Question
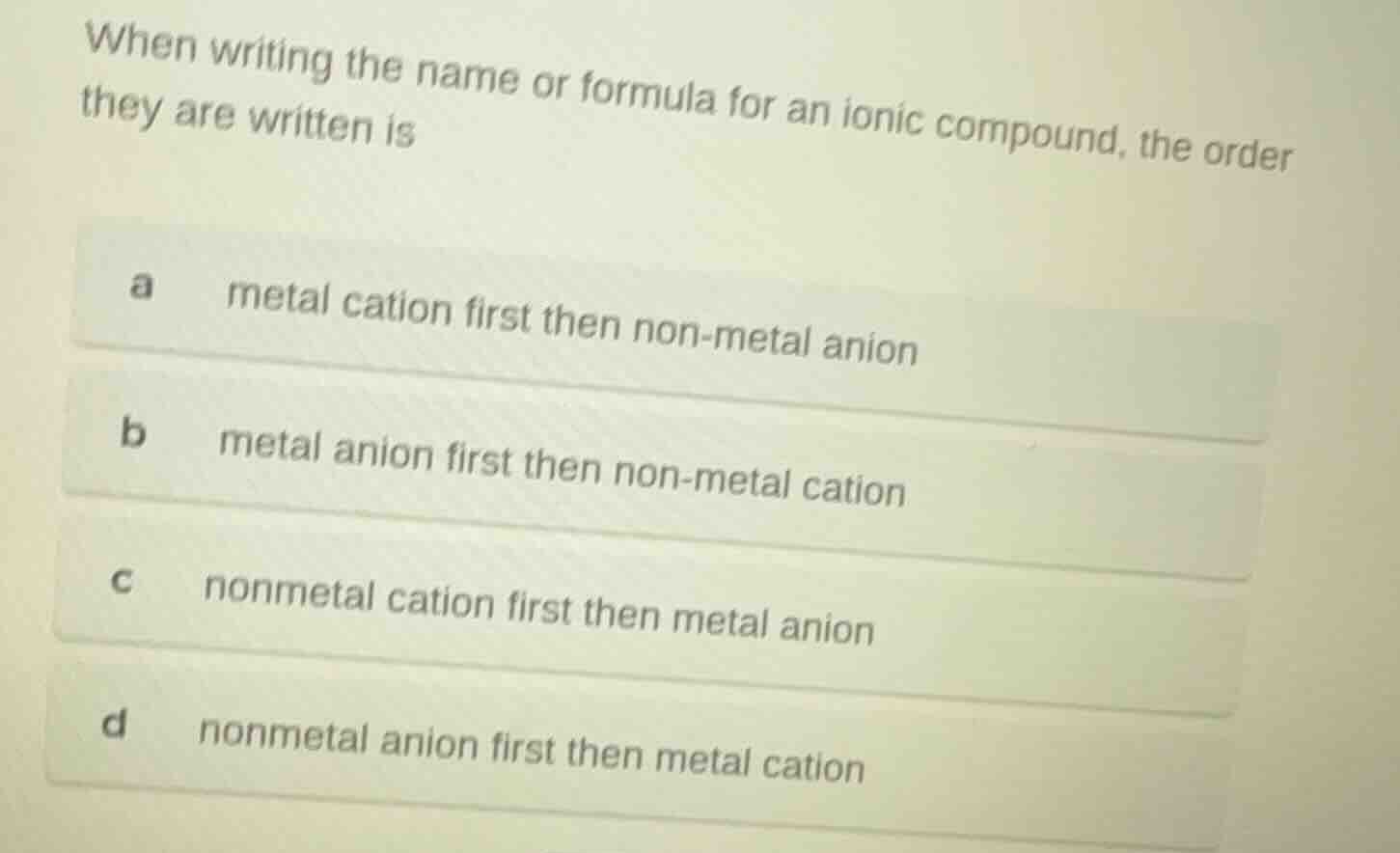
when writing the name or formula for an ionic compound, the order they are written is
a metal cation first then non - metal anion
b metal anion first then non - metal cation
c nonmetal cation first then metal anion
d nonmetal anion first then metal cation
Ionic compounds are formed from cations (positively charged ions) and anions (negatively charged ions). Metals typically form cations (metal cations), and non - metals typically form anions (non - metal anions). When naming or writing the formula for an ionic compound, the cation is written first, followed by the anion. So the metal cation (which is positive) comes first, and then the non - metal anion (which is negative). Option a follows this rule. Option b is incorrect because metals form cations, not anions in ionic compounds. Option c is incorrect as non - metals form anions, not cations, and the order is wrong. Option d has the anion first which is incorrect as the cation should be first.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a. metal cation first then non - metal anion