QUESTION IMAGE
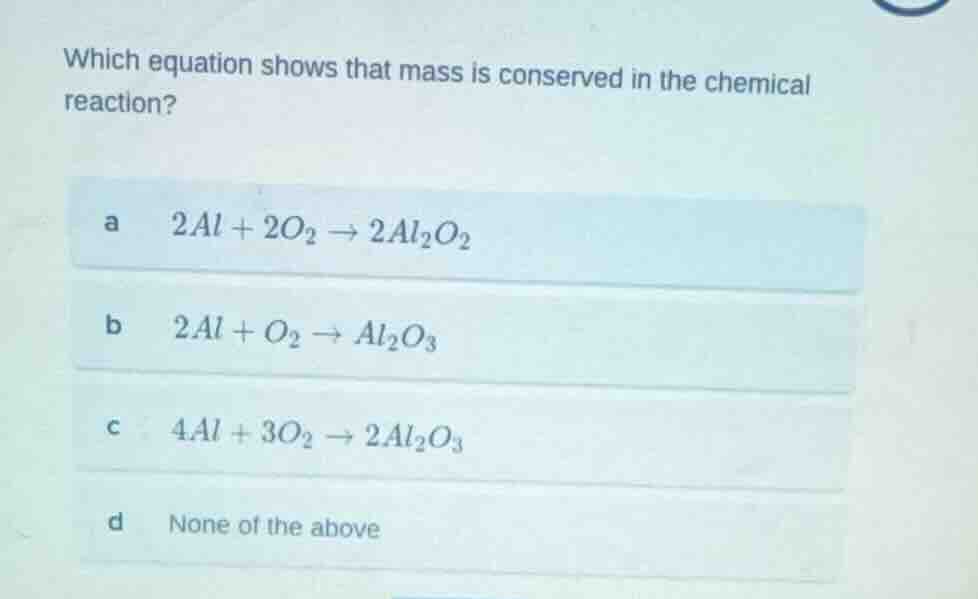
Question
which equation shows that mass is conserved in the chemical reaction?
a $2al + 2o_2 \
ightarrow 2al_2o_2$
b $2al + o_2 \
ightarrow al_2o_3$
c $4al + 3o_2 \
ightarrow 2al_2o_3$
d none of the above
Step1: Analyze Option a
Count atoms on left: Al = 2, O = \(2\times2 = 4\).
On right: Al = \(2\times2 = 4\), O = \(2\times2 = 4\)? Wait, formula \(Al_2O_2\) – Al: 2 per molecule, 2 molecules: 4. O: 2 per molecule, 2 molecules: 4. But left O is 4, left Al is 2. Not balanced (Al: 2 vs 4). So a is unbalanced.
Step2: Analyze Option b
Left: Al = 2, O = \(2\).
Right: \(Al_2O_3\) – Al = 2, O = 3. O atoms don’t match (2 vs 3). Unbalanced.
Step3: Analyze Option c
Left: Al = 4, O = \(3\times2 = 6\).
Right: \(2Al_2O_3\) – Al = \(2\times2 = 4\), O = \(2\times3 = 6\). Atoms of Al and O are equal on both sides. So this equation is balanced, meaning mass is conserved (Law of Conservation of Mass: atoms are neither created nor destroyed, so mass is conserved in a balanced chemical equation).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
c. \(4Al + 3O_2
ightarrow 2Al_2O_3\)