QUESTION IMAGE
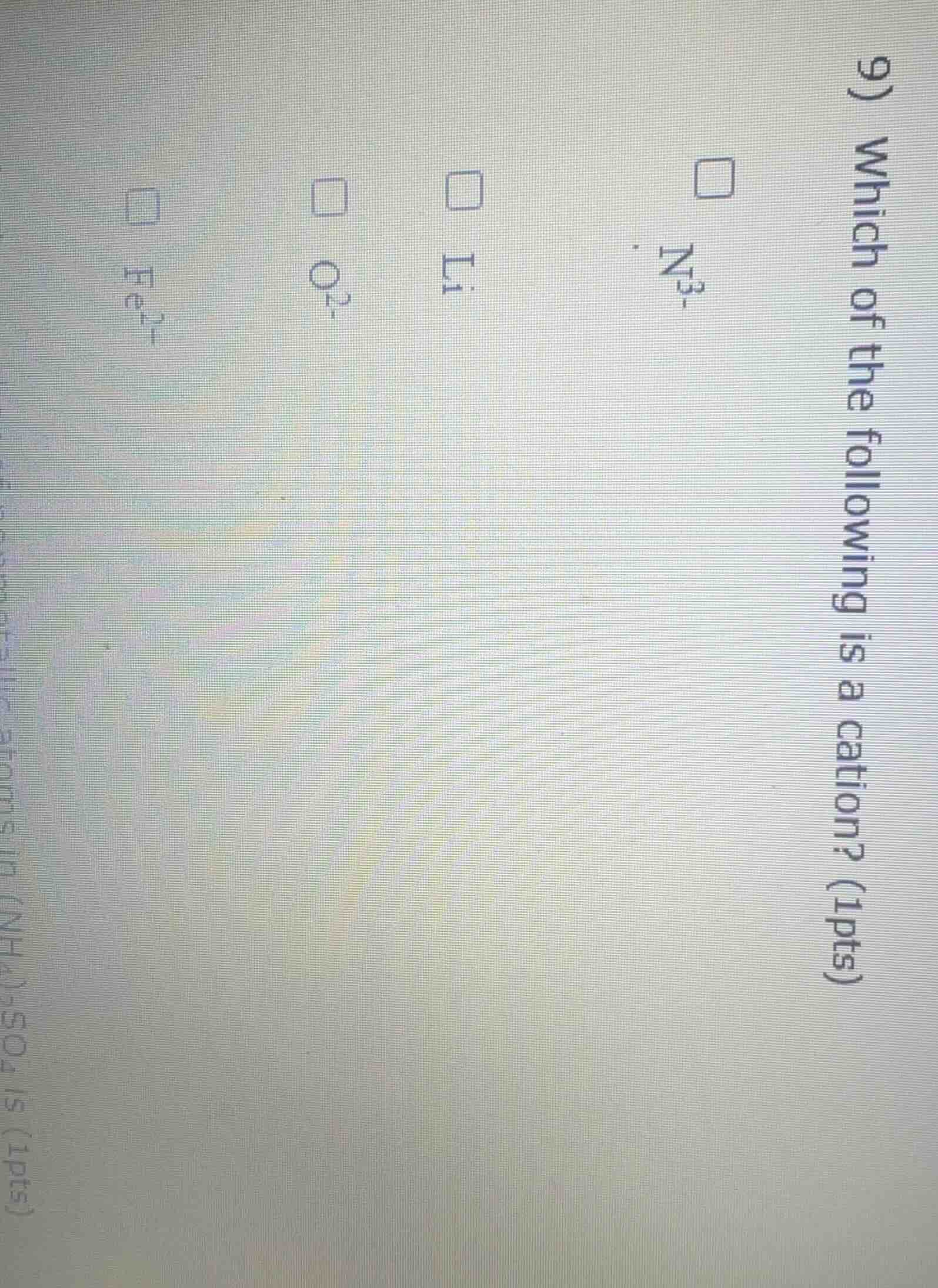
Question
- which of the following is a cation? (1pts) ☐ n³⁻ ☐ li⁺ ☐ o²⁻ ☐ fe²⁺
Brief Explanations
A cation is a positively charged ion. Analyze each option:
- $\text{N}^{3-}$: Has a 3- negative charge, an anion.
- $\text{Li}$: Neutral atom, no charge.
- $\text{O}^{2-}$: Has a 2- negative charge, an anion.
- $\text{Fe}^{2+}$: Has a 2+ positive charge, a cation.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{\text{Fe}^{2+}}$