QUESTION IMAGE
Question
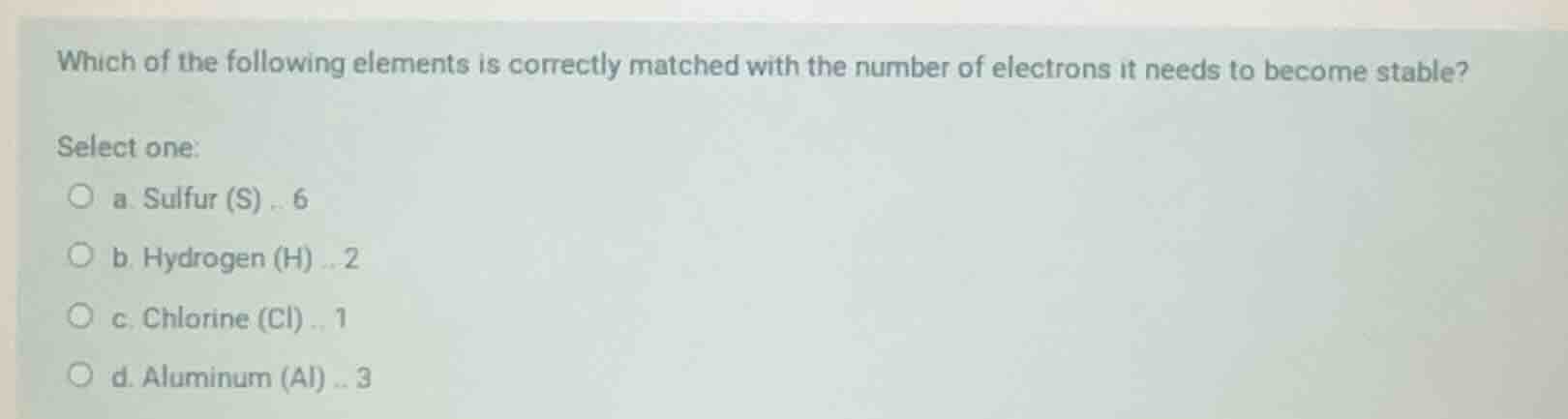
which of the following elements is correctly matched with the number of electrons it needs to become stable? select one: a. sulfur (s) .. 6 b. hydrogen (h) .. 2 c. chlorine (cl) .. 1 d. aluminum (al) .. 3
Step1: Analyze Sulfur's valence electrons
Sulfur has 6 valence electrons. To reach a stable octet, it needs $8-6=2$ electrons, not 6.
Step2: Analyze Hydrogen's valence electrons
Hydrogen has 1 valence electron. It only needs 1 more electron to fill its 1s orbital (stable duet), not 2.
Step3: Analyze Chlorine's valence electrons
Chlorine has 7 valence electrons. To reach a stable octet, it needs $8-7=1$ electron.
Step4: Analyze Aluminum's valence electrons
Aluminum has 3 valence electrons. It typically loses these 3 electrons to become stable (rather than gaining 5), so it does not need 3 electrons to become stable.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
c. Chlorine (Cl) .. 1