QUESTION IMAGE
Question
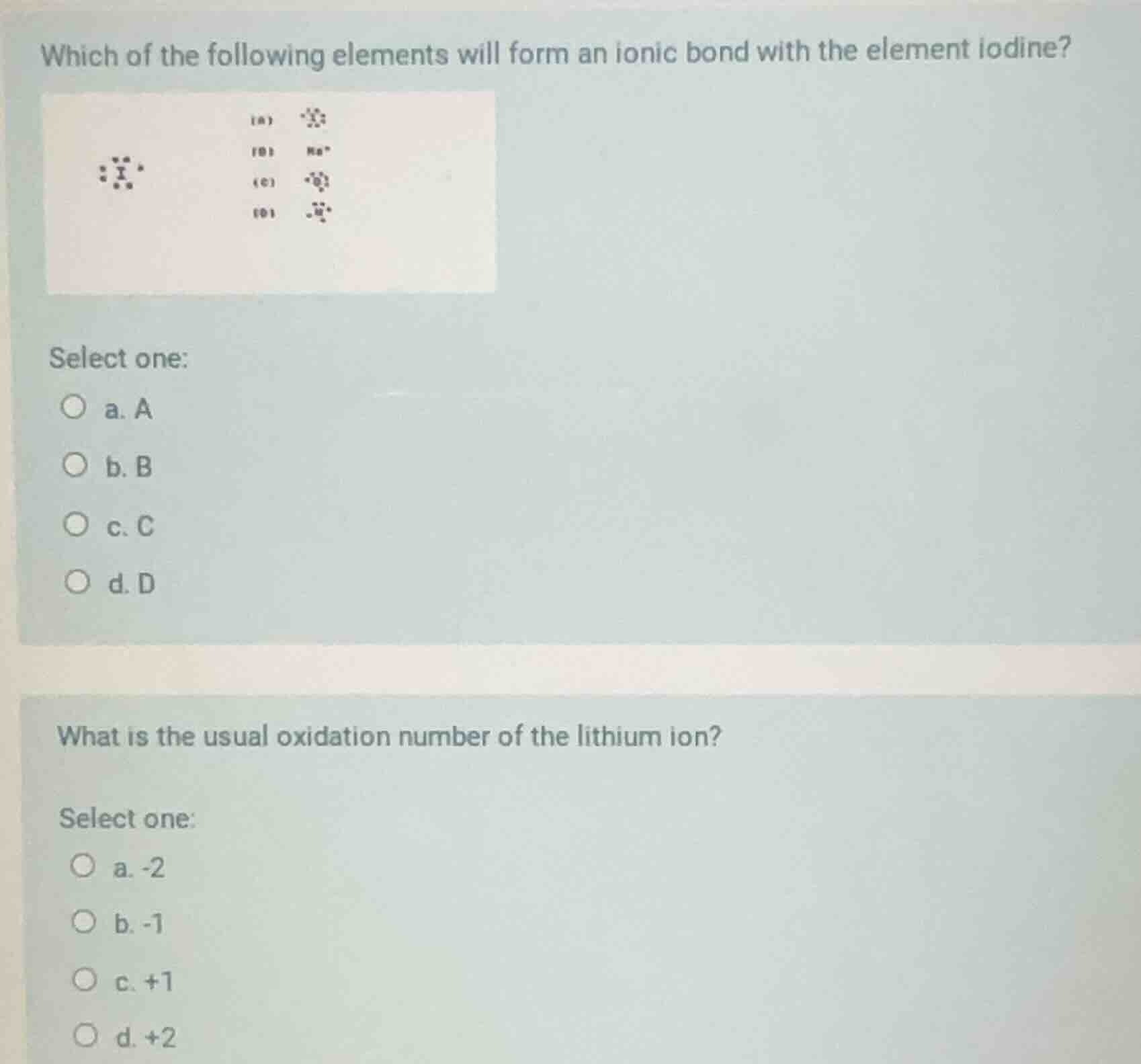
which of the following elements will form an ionic bond with the element iodine? (with some images of electron dot structures and ions) select one: a. a b. b c. c d. d what is the usual oxidation number of the lithium ion? select one: a. -2 b. -1 c. +1 d. +2
Step1: Analyze iodine's bonding behavior
Iodine (Group 17) is a nonmetal, gains 1 electron to form $\text{I}^-$. Ionic bonds form between metals (lose electrons) and nonmetals.
Step2: Identify the metal option
Option B is $\text{Na}^+$ (sodium, Group 1 metal), which loses 1 electron. $\text{Na}^+$ and $\text{I}^-$ form ionic $\text{NaI}$.
Step3: Determine lithium's oxidation number
Lithium is Group 1, has 1 valence electron, loses it to form $\text{Li}^+$, oxidation number = +1.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- b. B
- c. +1