QUESTION IMAGE
Question
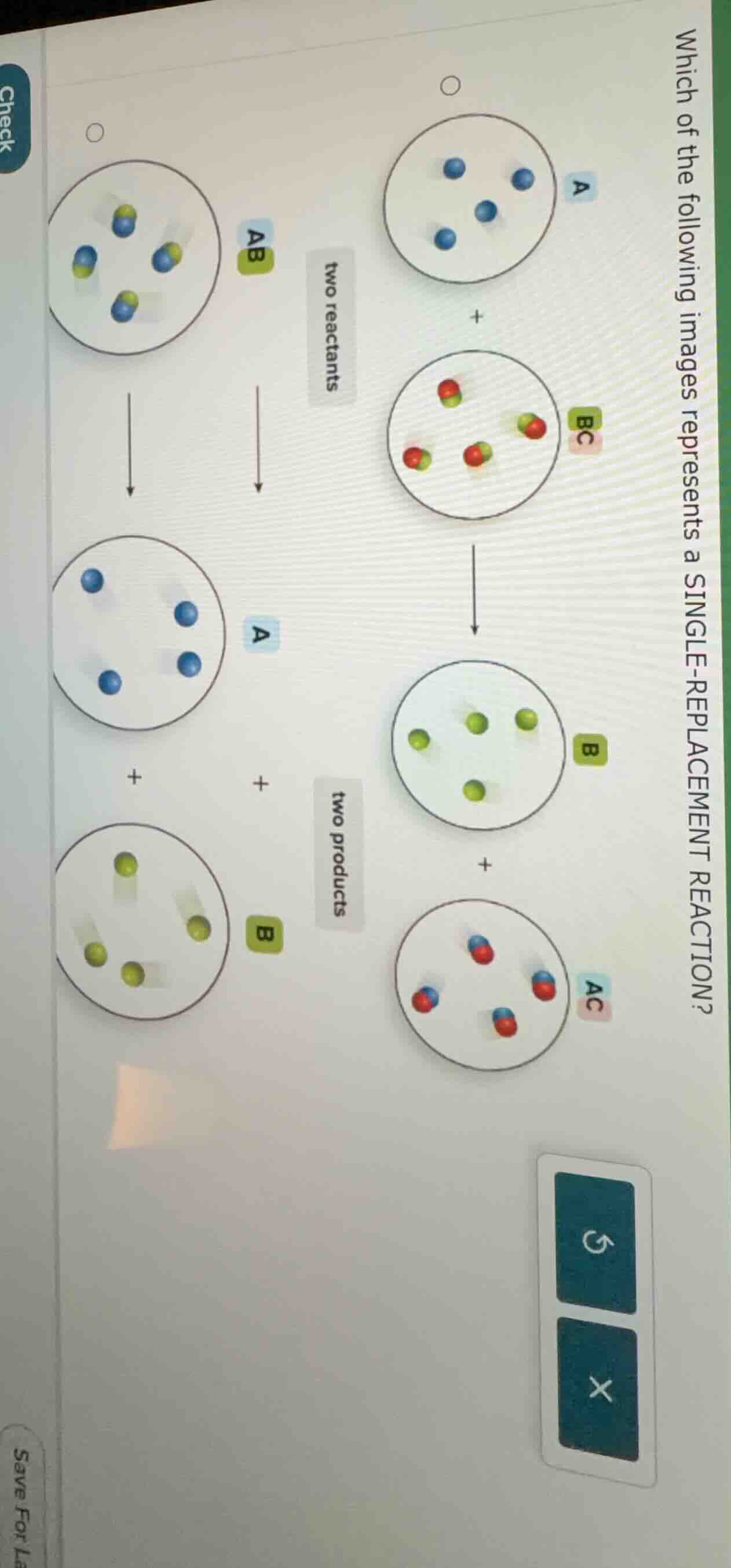
which of the following images represents a single - replacement reaction? two reactants two products
Brief Explanations
A single - replacement reaction has the general form \(A + BC
ightarrow AC + B\) (or \(Y+XZ
ightarrow YZ + X\)). Let's analyze each reaction:
- For the left - side reaction (with reactant \(AB\)): The reactants would be \(AB\) and another substance. But from the diagram, if we consider the reaction, it doesn't follow the single - replacement pattern. The products here are \(A\) and \(B\), which looks more like a decomposition reaction (if we assume \(AB
ightarrow A + B\)), not single - replacement.
- For the right - side reaction (with reactants \(A\) and \(BC\)): The reactants are an element \(A\) (the blue atoms) and a compound \(BC\) (the red - green molecules). The products are a compound \(AC\) (the red - blue molecules) and an element \(B\) (the green atoms). This follows the general form of a single - replacement reaction (\(A+BC
ightarrow AC + B\)).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The reaction with reactants \(A\) and \(BC\) (the right - hand side reaction in the diagram) represents a single - replacement reaction.